Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
(a) (i) Primary alcohols do no react appreciably with Lucas' reagent (HCl –ZnCl2) at room temperature.
(ii) Tertiary alcohol reacts immediately with Lucas 'reagent.
New answer posted
11 months agoContributor-Level 10
11.5 In this reaction, when propene reacts with the given reagent then the double bond of propene breaks down with charges on them. So, H+ gets placed on the carbon which already has two hydrogen atom and OH- gets substituted on center carbon because it has the more positive charge which attracts OH-. Thus we get propene-2-or as a
2. In this reaction, when Methyl ( 2-oxocyclohexyl) ethanoate reacts with the given reagent then the double bond between the oxygen atom and cyclohexyl gets breaks down, such that O has a negative charge and that particular carbon will have a positive charge on it. So, to neutralize it, H+ gets substitut
New answer posted
11 months agoContributor-Level 10
11.4 In the Grignard reagent reaction, the first step of the reaction is the nucleophilic addition of Grignard reagent to the carbonyl group to form an adduct, Hydrolysis of adduct results in the formation of alcohol.
Here, is the general reaction with Grignard reagent below:-
From here, it is clear that HCHO gives CH2OH groups, so R of Grignard reagent is the remaining part of given alcohols. Thus, select the suitable Grignard reagent by substituting the value of R. Now we can see the reaction given below:-
Methanal reacts with iso-propyl magnesium bromide, in presence of dry ether gives an additional compound. And this additional compou
New answer posted
11 months agoContributor-Level 10
11.3 (i) 3-chloroethyl-2-isopropylpentan-1-ol
(ii) 2,5-Dimethylhexane-1,3-diol
(iv) 3-Bromocyclohexanol
(v) Hex-1-en-3-ol
(vi) 2-Bromo-3-methylbut-2-en-1-ol
New answer posted
11 months agoContributor-Level 10
11.2 Allylic alcohol is an organic compound which has the structural formula CH2 = CHCH2OH. In other words, in these alcohols, the-OH group is attached to sp2 hybridized carbon next to the carbon-carbon double bond, that is to an allylic carbon. Therefore, in the above examples, the following are the allylic alcohols.
(ii) H2C = CH – CH2OH and
New answer posted
11 months agoContributor-Level 10
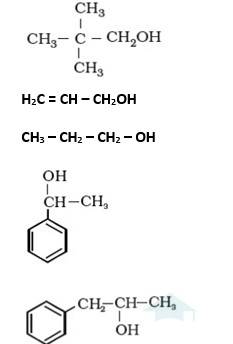
It is primary alcohol because carbon which carries the –OH group is only attached to one alkene group.
It is primary alcohol because the carbon which carries the –OH group is only attached to one propyl group.
It is secondary alcohol because the carbon which carries the –OH group is joined directly to methyl and benzene.
It is secondary alcohol because the carbon which carries the –OH group is joined directly to two different alkyl groups.
New answer posted
11 months agoContributor-Level 10
Ferromagnetism: The substances that are strongly attracted by a magnetic field are called ferromagnetic substances can be permanently magnetised even in the absence of a magnetic field. Some examples of ferromagnetic substances are iron, cobalt, nickel, gadolinium, and CrO2. In solid state, the metal ions of ferromagnetic substances are grouped together into small regions called domains and each domain acts as a tiny magnet. In an un-magnetised piece of a ferromagnetic substance, the domains are randomly-oriented and so, their magnetic moments get cancelled. However, when the substance is placed in a magnetic field, all the domains get
New answer posted
11 months agoContributor-Level 10
1.49 NaCl is doped with 10−3 mol % of SrCl2
100 moles of NaCl are doped with 0.001 moles of SrCl2
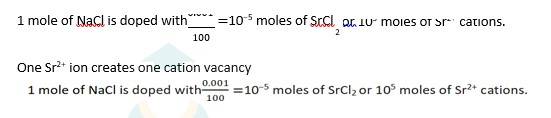
New answer posted
11 months agoContributor-Level 10
1.48 It is given that aluminum crystallises in a cubic closed packed structure.
Its metallic radius is 125 pm.
For cubic close-packed structure
a=2√2r=2√2*125=354 pm
Here, a is the edge length of the unit cell and r is the atomic radius.
(ii) Volume of one unit cell = a3 =(354 pm)3=4.4*10−23cm3(1 pm=10−10cm)
Number of unit cells in 1.00cm3= 1.00 cm3 / 4.4*10-23 cm3
= 2.27*1022
New answer posted
11 months agoContributor-Level 10
Schottky defect: Schottky defect is basically a vacancy defect shown by ionic solids. In this defect, an equal number of cations and anions are missing to maintain electrical neutrality. It decreases the density of a substance. Significant number of Schottky defects is present in ionic solids. For example, in NaCl, there are approximately 106 Schottky pairs per cm3 at room temperature. Ionic substances containing similar-sized cations and anions show this type of defect. For example: NaCl, KCl, CsCl, AgBr, etc.
Frenkel defect: Ionic solids containing large differences in the sizes of ions show this type of defect. When the smaller ion (
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers
