Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
If the bond formed between an enzyme and an inhibitor is a strong covalent bond and cannot be broken easily, then the enzyme is blocked permanently, The body then degrades the enzyme-inhibitor complex and synthesises the new enzyme.
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Receptor proteins are embedded in cell membranes in such a way that their small part possessing active sites projects out of the surface of the membrane and opens on the outside region of the cell membrane.
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
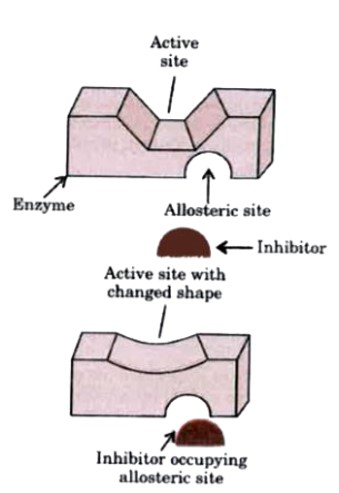
Some drugs do not bind to the enzyme's active site. These bind to a different site of enzyme which is called allosteric site. This bonding of inhibitor at allosteric site changes the shape of the active site in such a way that substrate cannot recognise it. As a result, the affinity of the substrate for the active site is reduced.
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Sodium salts of benzoic acid, sorbic acid and propanoic acid, etc., are used as food preservatives.
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Since aspartame is unstable and decomposes at cooking temperature, therefore, it is used as a sweetening agent in cold foods and soft drinks.
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
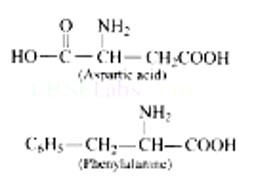
Aspartic acid and phenylalanine are two a-amino acids which form the methyl ester of dipeptide and named as aspartame (an artificial sweetener) which is 100 times more sweet than cane sugar.
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Sucralose is trichloro derivative of sucrose. It is about 600 times sweeter than sucrose. However, it neither provides calories nor causes tooth decay.
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
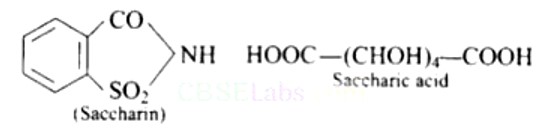
Saccharin (o-sulpho benzoicimide) is an artificial sweetener, whereas saccharic acid (dicarboxylic acid) is obtained by the oxidation of glucose with cone. HNO3 or by bacterial oxidation.
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Table salt, sugar vegetable oils and sodium benzoate, etc. are used as preservatives. These do not allow moisture and air to enter the material and hence, bacteria cannot thrive on them. Therefore, pickles do not get spoiled for months.
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
In low calorie drinks, some artificial sweetening agents (like aspartame, alitame, sucralose, saccharin etc.) are present which are often many hundred times sweeter than sugar but do not metabolise and hence, do not produce any energy.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers