Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
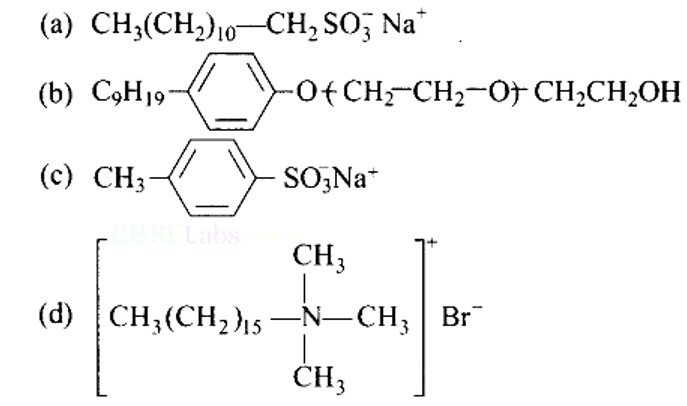
This is a multiple choice answer as classified in NCERT Exemplar
(b) Liquid dishwashing detergents are non-ionic detergents.
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (B)
Hoffmann bromamide reaction- it is also called degradation reaction as in this reaction primary amide group is treated with halogen first (Br) then the halogen-substituted amide product is converted to a primary amine with the release of carbon dioxide gas.
New answer posted
9 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(c) Glycerol is added to shaving soap to prevent rapid drying while to enhance the leathering property of soap, a gum called rosin is added to them. It forms sodium rosinate which lathers well.
New answer posted
9 months agoContributor-Level 10
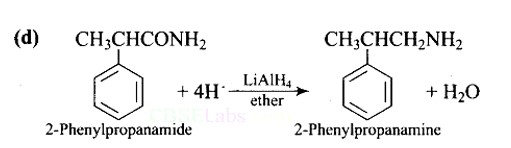
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (D)
Lithium aluminium hydride in ether is a strong reducing agent that donates its hydride ion to any C=O containing functional groups into corresponding reduced compounds. Here, the amide group is converted to an amine functional group.
LiAlH4 in ether is the best reagent for converting 2-phenylpropanamide to 2-phenylpropanamine.
New answer posted
9 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(b) Shaving soaps contain glycerol to prevent rapid drying. A gum called rosin is added in these soaps which forms sodium rosinate which enhances lathering property of soap.
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: ( C)
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (D)
Gabriel synthesis reaction:
Gabriel's synthesis is a method for producing primary amines. When phthalimide is treated with ethanolic potassium hydroxide, it forms a potassium salt of phthalimide, which when heated with an alkyl halide and then alkaline hydrolyzed yields the corresponding primary amine. This method cannot produce aromatic primary amines because aryl halides do not undergo nucleophilic substitution with the anion formed by phthalimide.
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (C)
Potassium cyanide, as cyanide on reduction with sodium metal in alcohol, produces amine with increased carbon atoms.
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (B)
Lithium aluminum hydride in ether is a strong reducing agent that donates its H? hydride ion to any C=O containing a functional group. In addition to LiAlH4 to aryl nitro compounds, no reaction will be observed, the desired products of amines will not be produced, rather it will form diazobenzene products.
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (C)
SN1 reaction: A nucleophilic reaction that occurs in two steps, first is the bond-breaking step and the second is the production of the carbocation. The stability of carbocation formed in the second step determines the rate of reactivity of reactant toward SN1 reaction. Here, C6H5CH2Br,
In the process of ionization, removal of bromine, a stable Benzyl carbocation is produced. Therefore, it is best suited for reaction through the SN1 mechanism.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers