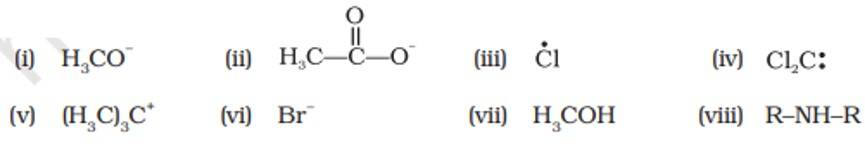Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
(a) Fully filled and half-filled orbitals have extra stability (that is, lower energy). Thus, p3, p6, d5, d10, f 7, f14 etc. configurations, which are either half-filled or filled, are more stable. Chromium and copper therefore adopt the d5 and d10 configuration.
New answer posted
9 months agoContributor-Level 10
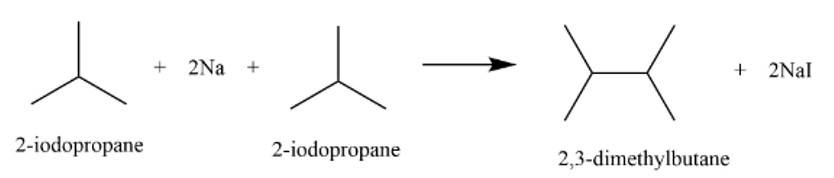
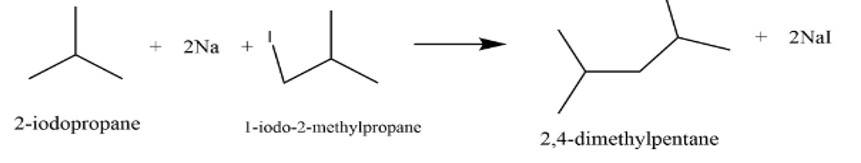
This is a short answer type question as classified in NCERT Exemplar
It is a Wurtz reaction where alkane is formed.
New answer posted
9 months agoContributor-Level 10
(c) The path of an electron in an atom can never be determined or known accurately. That is why there is only a probability of finding the electron at different points in an atom.
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The structure of 2-methylbutane is
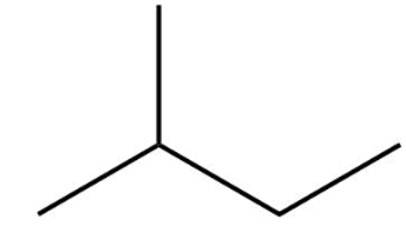
2-methylbutane
Total number | Total amount of monochlorinated Product | % of monochlorinated product | |
1° | 9 | 9 | 41.7 |
2° | 2 | 7.6 | 35.2 |
3° | 1 | 5 | 23.1 |
New answer posted
9 months agoContributor-Level 10
(a) Classical mechanics applies to the macro world and hence fails when it is applied to the micro world. In the micro world, quantum mechanics incorporates all the principles like the uncertainty principle, dual behaviour etc.
New answer posted
9 months agoContributor-Level 10
(d) Rainbow is a continuous spectrum.
Assertion is a wrong statement but Reason is a correct one. Rainbow is a continuous spectrum.
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Electrophiles | Nucleophiles |
(iii), (iv), (v) | (i), (ii), (vi), (vii), (viii) |
New answer posted
9 months agoContributor-Level 10
(a) For n = 4, No. of sub-shells = (l = 0, l = 1, l = 2, l = 3) = 4.
(b) Total number of orbitals which can be present = n2 = 42 = 16.
Each orbital can have an electron with ms = – 1/2. Total no. of electrons with m, = – 1/2 is 16.
New answer posted
9 months agoContributor-Level 10
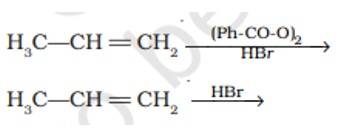
This is a short answer type question as classified in NCERT Exemplar
In presence of (Ph-CO-O)2it lead to CH3-CH2-CH2Br as the reaction undergo by free radical mechanism
However in absence of (Ph-CO-O)2 the reaction undergo by carbocation intermediate thus lead to give CH3-CHBr-CH3
New answer posted
9 months agoContributor-Level 10
(a) P (Z=15) : [Ne]103s23p3 No. of unpaired electrons = 3
(b) Si (Z=14) : [Ne]103s23p2 No. of unpaired electrons = 2
(c) Cr (Z=24): [Ar]184s13d5 No. of unpaired electrons = 6
(d) Fe (Z=26): [Ar]184s23d6 No. of unpaired electrons = 4
(e) Kr (Z=36) : [Ar]184s23d104p6 No. of unpaired electrons = Nil.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers