Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(iv) 0.016
Explanation: Apply the relation : M1V1= M2V2
Given: M1=0.02M, V1=4L, M2=? V2=5L
Therefore, 0.02*4L=M2*5L
M2=0.08/5
=0.016 M
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(iv) A–B interactions are weaker than those between A–A or B–B.
Explanation: (i) At a given composition, the solutions that demonstrate a big positive divergence from Rault's law form a minimum boiling azeotrope.
(ii) When Rault's law is deviated positively, A-B interactions are weaker than A-A or B-B interactions.
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
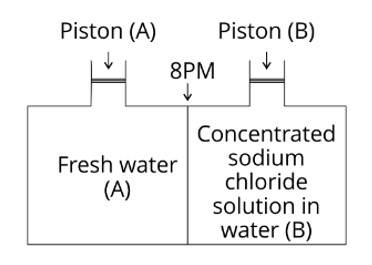
(i) Vapour pressure in container (A) is more than that in container (B).
Explanation: Due to the fleeing inclinations of water molecules from the liquid's surface, the vapour pressure rises. The vapour pressure increases as the number of molecules on the liquid's surface increases. Because only water molecules are present at the surface of beaker A, it has a higher vapour pressure. However, a fraction of the surface area of the solution in beaker B containing NaCl solution is occupied by NaCl molecules, which are non-volatile and have no tendency to escape. As a resul
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(ii) Solution (A) will follow Raoult's law.
Explanation: A-A and B-B intermolecular interactions should be almost identical to A-B type interactions in an ideal solution.
New question posted
10 months agoNew question posted
10 months agoNew answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(ii) Water will move from side (B) to side (A) if a pressure greater than osmotic pressure is applied on piston (B).
Explanation: Due to reverse osmosis, water will travel from side (B) to side (A) if a pressure greater than osmotic pressure is applied to the piston (B).
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(ii) Greater for gases with lower solubility.
Explanation : Because of the mathematical relationship, the value of Henry's constant KH is bigger for gases with lesser solubility-
p= kH x
kH=p/x
Where, KH represents Henry's constant, p is partial pressure of the gas in vapour phase, and x denotes mole fraction of the gas in solution. Thus KH is inversely proportional to mole fraction of gas in solution (representing its solubility)
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(i) Increases with increase in temperature.
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(ii) In reverse osmosis, solvent molecules move through a semipermeable membrane from a region of lower concentration of solute to a region of higher concentration.
Explanation:
Solvent molecules pass through a semipermeable membrane in reverse osmosis from an area of higher solute concentration to a region of lower concentration, hence the supplied assertion at (ii) is untrue.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers