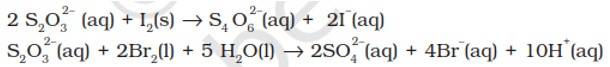Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
5.25. (a) Other than attractive forces, molecules also exert repulsive forces on one another. When two molecules are brought into close contact with each other, the repulsion between the electron clouds and that between the nuclei of two molecules comes into play. Magnitude of the repulsion rises very rapidly as the distance separating the molecules decreases. This is the reason that liquids and solids are hard to compress. In these states molecules are already in close contact; therefore, they resist further compression; as that would result in the increase of repulsive interactions.
New answer posted
11 months agoContributor-Level 10
(a) The balanced half reaction equations are:
Oxidation half equation:
I− (aq) → I2 (s) - (i)
Reduction half reaction equation:
MnO4− (aq) → MnO2 (aq) - (ii)
Balance I atoms and charges in the oxidation half reaction.
2I− (aq) → I2 (s) + 2e−
In the reduction half reaction, the oxidation number of Mn changes from +7 to +4. Hence, add 3 electrons to reactant side of the reaction.
MnO4− (aq) + 3e−→ MnO2 (aq)
Balance charge in the reduction half reaction by adding 4 hydroxide ions to product side.
MnO4− (aq) + 3e− → MnO2 (aq)+4OH−
To balance O atoms, add 2 water molecules to reactant s
New answer posted
11 months agoContributor-Level 10
5.24. (b) Viscosity of liquids decreases as the temperature rises because at high temperature molecules have high kinetic energy and can overcome the intermolecular forces to slip past one another between the layers.
New answer posted
11 months agoContributor-Level 10
5.23. (a) The dipole-dipole interaction between two HCl molecules is stronger than the London forces but is weaker than ion-ion interaction because only partial charges are involved.
New answer posted
11 months agoContributor-Level 10
Reactions (a) and (b) indicate that H3PO2 (hypophosphorous acid) is a reducing agent and thus reduces both AgNO3 and CuSO4 to Ag and Cu respectively. Conversely, both AgNO3 and CuSO4 act as oxidising agent and thus oxidise H3PO2to H3PO4 (orthophosphoric acid) Reaction (c) suggests that [Ag (NH3)2]+ oxidises C6H5CHO (benzaldehyde) to C6H5COO– (benzoate ion) but reaction (d) indicates that Cu2+ ions cannot oxidise C6H5CHO to C6H5COO–. Therefore, from the above reactions, we conclude that Ag+ ion is a strong deoxidising agent than Cu2+ ion.
New answer posted
11 months agoContributor-Level 10
XeO64−? oxidizes F− and F− reduces XeO64−?
Hence, the given reaction occurs.
The oxidation number of Xe decreases from +8 to +6. The oxidation number of F increases from -1 to 0.
Thus, Na4? XeO6? is a stronger oxidising agent than F−.
New answer posted
11 months agoContributor-Level 10
Fluorine oxidizes chloride ion to chlorine, bromide ion to bromine and iodide ion to iodine respectively.
F2? + 2Cl− → 2F− + Cl2?
F2? + 2Br− → 2F− + Br2?
F2? + 2I− → 2F− + I2?
Chlorine oxidizes bromide ion to bromine and iodide ion to iodine.
Cl2? + Br− → 2Cl− + Br2?
Cl2? + I− → 2Cl− + I2?
Bromine oxidizes iodide ion to iodine.
Br2? + I− → 2Br− + I2?
But bromine and chlorine cannot oxidize fluoride to fluorine. Hence, fluorine is the best oxidizing agent amongst the halogens. The decreasing order of the oxidizing power of halogens is F2? >Cl2? >Br2? >I2?
HI and HBr can reduce sulphuric acid
New answer posted
11 months agoContributor-Level 10
The average O.N. of S in S2O32- is +2 while in S4O62- it is + 2.5. The O.N. of S in SO42- is +6. Since Br2 is a stronger oxidising agent than I2, it oxidises S of S2O32- to a higher oxidation state of +6 and hence forms SO42- ion. I2, however, being weaker oxidising agent oxidises S of S2O32- ion to a lower oxidation of +2.5 in S4O62- ion. It is because of this reason that thiosulphate reacts differently with Br2 and I2.
New answer posted
11 months agoContributor-Level 10
11.86

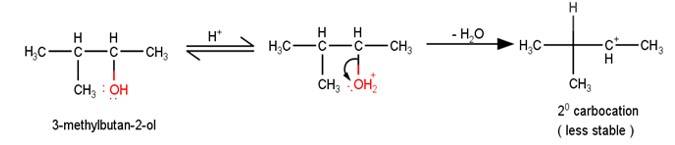
The first step in the mechanism of th e given reaction is protonation of the alcohol followed by loss of water to give a 20 carbocation.
e given reaction is protonation of the alcohol followed by loss of water to give a 20 carbocation.
2. The next step is a rearrangement of the 20 carbocations formed in the above step is less stable it rearranges by a 1,2-hydride shift to form more stable 3° carbocations.
New answer posted
11 months agoContributor-Level 10
Substance oxidised | Substance reduced | Oxidising agent | Reducing agent |
(a) C6H12O6 | AgBr | C6H12O6 | |
(b) HCHO | [Ag (NH3)2]+ | [Ag (NH3)2]+ | HCHO |
(c) HCHO | Cu2+ | HCHO | |
(d) N2H4 | H2O2 | N2H4 | |
(e) Pb | PbO2 | Pb |
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers