Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
5.22. Higher the critical temperature, more easily the gas can be liquefied, i.e., greater are the intermolecular forces of attraction. Hence, CO2 has stronger intermolecular forces than CH4.
New answer posted
11 months agoContributor-Level 10
(a) Toluene can be oxidised to benzoic acid in acidic, basic and neutral media according to the following redox equations:
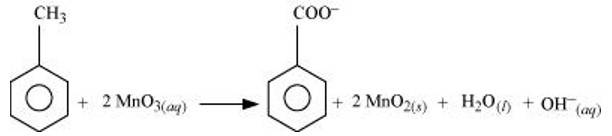
In the laboratory, benzoic acid is usually prepared by alcoholic KMnO4 oxidation of toluene. However, in industry alcoholic KMnO4 is preferred over acidic or alkaline KMnO4 because of the following reasons:
(i) The cost of adding an acid or the base is avoided because in the neutral medium, the base (OH- ions) are produced in the reaction itself.
(ii) Since reactions occur faster in homogeneous medium than in heterogeneous medium, therefore, alcohol helps in mixing the two reactants, i.e., KMnO4
New answer posted
11 months agoContributor-Level 10
11.85
We know that the addition and elimination reactions are opposite of each other.Hence, for solving the above questions our approach should be to first dehydrate a suitable alcohol to give either a single alkene or a mixture of an alkene, if we obtain a mixture of alkene then we would have to detect which of the alkene will give us the desired alcohol. Wherever required the acid- catalyzed addition of water to alkenes will follow Markovnikov's rule.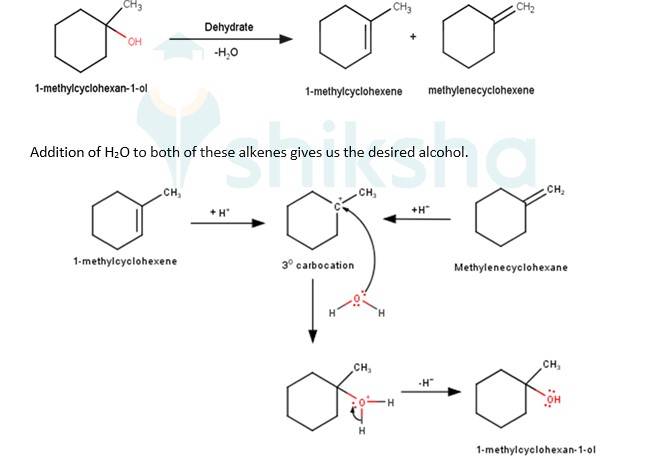
New answer posted
11 months agoContributor-Level 10
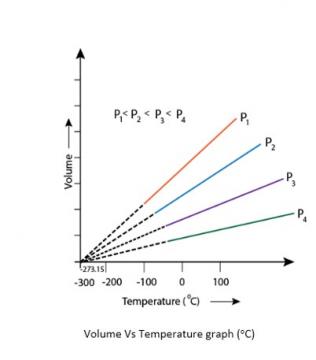
5.21. At -273°C, volume of the gas becomes equal to zero, i.e., the gas ceases to exist.
New answer posted
11 months agoContributor-Level 10
The three examples are:
(i) When excess P4? (reducing agent) reacts with F2? (oxidizing agent), PF3? is produced in which P has +3 oxidation number.
P4? (excess) + F2? → PF3?
But if fluorine is in excess, PF5? is formed in which P has oxidation number of +5.
P4? ? + F2? (excess) → PF5?
(ii) Oxidizing agent is oxy
New answer posted
11 months agoContributor-Level 10
5.20. SI unit of pressure, P = Nm-2
SI unit of volume = m3
Si unit of temperature, T = K
SI unit of number of moles, n = mol
Thus, SI unit of pV2T2/n = (Nm-2) (m3)2 (K) 2mol = Nm4K2mol-1
New answer posted
11 months agoContributor-Level 10
In AgF2, oxidation state of Ag is +2 which is very unstable. Therefore, it quickly accepts an electron to form the more stable +1 oxidation state.
Ag2+ + e– →Ag+
Therefore, AgF2, if formed, will act as a strong oxidising agent.
New answer posted
11 months agoContributor-Level 10
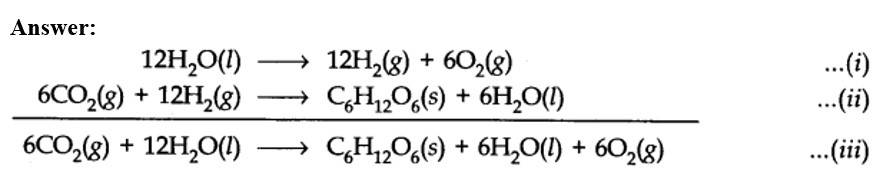
(a) Therefore, it is more appropriate to write the equation for photosynthesis as (iii) because it emphasises that 12H2O are used per molecule of carbohydrate formed and 6H2O are produced during the process.
(b) The purpose of writing O2 two times suggests that O2 is being obtained from each of the two reactants.
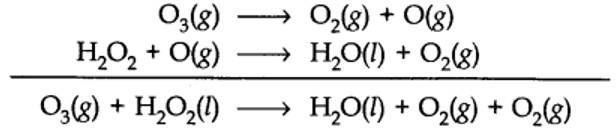
(a) or by using H2O218 or O318in reaction (b).
New answer posted
11 months agoContributor-Level 10
(i) In SO2, O.N. of S is +4. In principle, S can have a minimum O.N. of -2 and maximum of +6. Therefore, S in SO2 can either decrease or increase its O.N. and hence can act both as an oxidising as well as a reducing agent.
(ii) In H2O2, the O.N. of O is -1. In principle, O can have a minimum O.N. of -2 and maximum of zero (+1 is possible in O2F2 and +2 in OF2). Therefore, O in H2O2 can either decrease its O.N. from -1 to -2 or can increase its O.N. from -1 to zero. Therefore, H2O2 acts both as an oxidising as well as a reducing agent.
(iii) In O3, the O.N. of O is zero. It can only decrease its O.N. from zero to -1 or -
New answer posted
11 months agoContributor-Level 10
Substance | Oxidation number of C | Substance | Oxidation number of N |
CH2? Cl2? | 0 | N2? | 0 |
FC≡CF | +1 | N2? O | +1 |
HC≡CH | -1 | N2? H2? | -1 |
CHCl3? , CO | +2 | NO | +2 |
CH3? Cl | -2 | N2? H4? | -2 |
Cl3? C−CCl3? | +3 | N2? O3? | +3 |
H3? C−CH3? | -3 | NH3? | -3 |
CCl4? , CO2? | +4 | NO2? | +4 |
CH4? | -4 | N2? O5? | +5 |
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers
