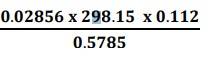Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
1.37. (c) Equal moles of different substances contain same number of constituent particles but equal weights of different substances do not contain the same number of constituent particles due to the difference in their molar mass.
New answer posted
11 months agoContributor-Level 10
(d) The ionization enthalpies of the alkali metals are considerably low and decrease down the group from Li to Cs. This is because the effect of increasing size outweighs the increasing nuclear charge, and the outermost electron is very well screened from the nuclear charge.
New question posted
11 months agoNew question posted
11 months agoNew answer posted
11 months agoContributor-Level 10
1.36. The molar mass of MnO2is 87g and the molar mass of HCl is 36.5 g.
According to the equation, 4 moles of HCl (i.e. 4 x 36.5g = 146 g) reacts with 1 mol of MnO2 (87g).
So, for 5.0 g of MnO2 will react with:
= 5 x146/87 = 8.40 g of MnO2
Therefore, 8.4 g of HCl will react with 5 g of MnO2.
New answer posted
11 months agoContributor-Level 10
1.35. Step 1: 0.75 M HCl means 0.75 mol per 1000 mL or 0.75 x 36.5 g in 1000 mL.
i.e. 1000 mL of 0.75 M HCl contains 0.75 x 36.5 g HCl
Therefore, 25 mL of 0.75 M HCl contains 0.75 x 36.5 x 25/ 1000 g = 0.6844 g
Step 2: To calculate mass of CaCO3reacting completely with 0.6844 g of HCl
CaCO3 (s) + 2HC1 (aq)———>CaCl2 (aq) +CO2 (g) + H2O
2 mol of HCl, i.e., 2 x 36.5 g = 73 g HCl react completely with CaC03 = 1 mol = 100 g
Therefore, 0.6844 g HCl will react completely with CaCO3 = 100/73 x 0.6844 g = 0.938 g
New answer posted
11 months agoContributor-Level 10
1.34. (i) 1 mole (44 g) of CO2 will have 12 g carbon.
So, 3.38 g of CO2 will have carbon = 12g/44g * 3.38
= 0.9217 g
18 g of water will have 2 g of hydrogen.
So, 0.690 g of water contain hydrogen = 2g/18g * 0.6902g
= 0.0767 g
Since carbon and hydrogen are the only constituents of the compound, the total mass of the compound is:
= 0.9217 g + 0.0767 g
=0.9984 g
So, the percentage of Carbon in the compound = 0.9217/0.9984 * 100 = 92.32%
Now, percentage of Hydrogen in the compound = 0.0767/0.9984 * 100 = 7.68%
Moles of carbon in the compound = 92.32/12=7.69
Moles of hydrogen in the compound = 7.68/1=7.68
Since, we ha
New answer posted
11 months agoContributor-Level 10
1.33. (i) 52 moles of Ar
Ans:1 mole of Ar = 6.022 * 1023 atoms of Ar
Therefore, 52 mole of Ar = 52 * 6.022 * 1023 atoms of Ar= 3.131 * 1025 atoms of Ar
(ii) 52 u of He
Ans:1 atom of He = 4u of the He
Or, 4 u of He = 1 atom of He
So, 52 u of He = 52/4 atom of He = 13 atoms of He.
(iii) 52 g of He
Ans:4g of He = 6.022 * 1023 atoms of He
So, 52g of He = 6.022 * 1023 * 52/4 atoms of He
= 7.8286 * 1024 atoms of He
New answer posted
11 months agoContributor-Level 10
1.32. Molar mass of argon is
= [ (35.96755 * 0.337/100)+ (37.96272 * 0.063/100)+ (39.9624 * 99.60/100)]g mol-l
= [0.121+0.024+39.802] g mol-l
= 39.947 g mol-l
So, the molar mass of argon is 39.947 g/ mol.
New answer posted
11 months agoContributor-Level 10
1.31. First we need to find the least precise number to find the significant figures.
(i) Least precise number of the calculation![]() is 0.112
is 0.112
Therefore, number of significant figures in the answer = Number of significant figures in the least precise number, i.e. 3
(ii) Least precise number of calculations = 5.364
Therefore, number of significant figures in the answer will be = Number of significant figures in 5.364 = 4
(iii) 0.0125+0.7864+0.0215
Since the least number of decimal places in each term is four, the number of significant figures in the answer will also be 4.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers