Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoThe nature of charge on resulting colloidal particles when FeCl3 is added to excess of hot water is:
New answer posted
8 months agoContributor-Level 10
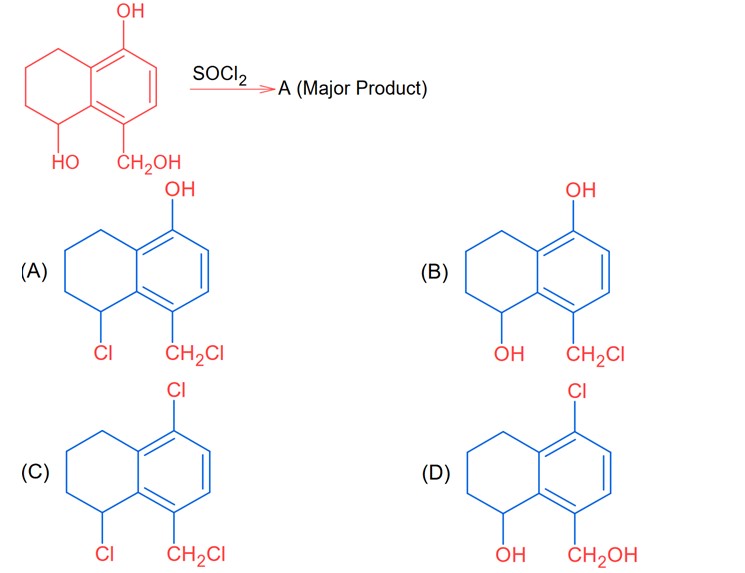
Since SOCl2 is used to covert aliphatic (R-OH) into chlorides. It will not react with aromatic alcohol
New answer posted
8 months agoContributor-Level 10
Siderite – FeCO3 (ore of iron)
Calamine – ZnCO3 (ore of zinc)
Malachite – CuCO3.Cu (OH)2 (ore of copper)
Cryolite – Na3AlF6 (ore of aluminium)
New answer posted
8 months agoContributor-Level 10
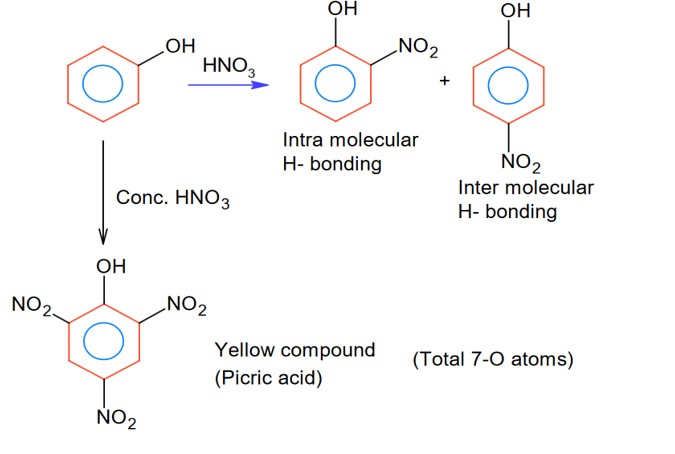
Intermolecular H- bonding and intra-molecular H- bonding producing compound may be the phenol derivatives.
New answer posted
8 months agoContributor-Level 10
Most basic oxide V2O3
Here V has +3 O.S. Hence V+3 ->
two unpaired e- in d- subshell
New answer posted
8 months agoContributor-Level 10
Volume of H2 adsorbed =
Therefore volume of gas adsorbed per gram of the adsorbent =
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers