Physics
Get insights from 5.6k questions on Physics, answered by students, alumni, and experts. You may also ask and answer any question you like about Physics
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
For a given stress, the strain in rubber is more than it is in steel, hence the Young's modulus of rubber is lesser than in steel. So the statement is False.
Shear modulus is the ratio of the applied stress to the change in the shape of a body. The stretching of a coil changes its shape. Hence, shear modulus of elasticity is involved in this process.
= 2.2
New answer posted
11 months agoContributor-Level 10
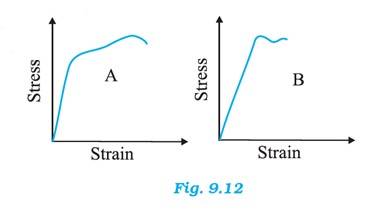
Material A has greater Young's modulus.
Material A is the strongest as it can withstand more strain than material B without fracture.
New answer posted
11 months agoContributor-Level 10
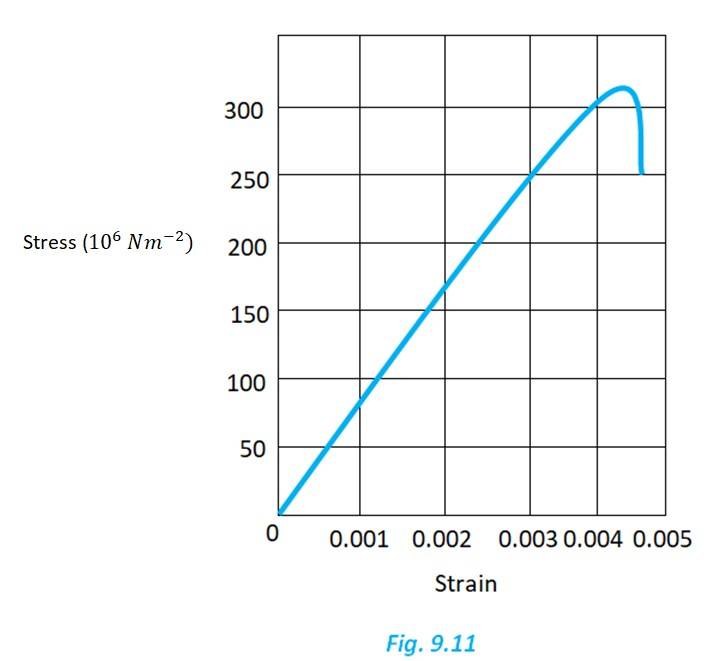
From the given graph, for the value stress 150 N/ , the strain is 0.002
Young's modulus = = 7.5 N/
Yield strength is the maxium strength the material can withstand in elastic limit. From the graph, the yield strength is 300 or 3
New answer posted
11 months agoContributor-Level 10
Length of the steel wire, = 4.7 m
Area of cross-section of the steel wire, = 3.0 m2
Length of the copper wire, = 3.5 m
Area of cross-section of the copper wire, = 4.0 m2
Change in length,
Let the force applied = F
Young's modulus in steel wire,
= ….(1)
Young's modulus in copper wire,
= …….(2)
The ratio of Young's modulus
= = = =
New answer posted
11 months agoContributor-Level 10
11.20 Potential difference across evacuated tube, V = 500 V
Specific charge of electron, e/m = 1.76 C/kg
The speed of each emitted electron is given by the relation of kinetic energy as
= =
= = = 13.27 m/s
Therefore, the speed of each emitted electron is 13.27 m/s
Collector potential, V = 10 MV = 10 V
The speed is given by = = 1.876 m/s
This is not possible nothing can move faster than the light. In the above formula
= can only be used in the non-relativistic limit, i.e. v << c
For very high speed problems
New answer posted
11 months agoContributor-Level 10
11.19 Temperature of the Nitrogen molecule, T = 300 K
Atomic mass of nitrogen = 14.0076 u
Hence, mass of Nitrogen molecule, m = 2 14.0076 u = 28.0152 u
We know, 1 u = 1.66 kg
So, m = 28.0152 1.66 kg = 4.65 kg
Planck's constant, h = 6.626 Js
Boltzmann constant, k = 1.38 kg
We have the expression that relates to mean kinetic energy ( of the nitrogen molecule with root mean square speed ( ) as:
=
= = 516.814 m/s
De Broglie wavelength of the nitrogen molecule
= = 2.76 m = 0.0276 nm
Therefore,
New answer posted
11 months agoContributor-Level 10
11.18 The momentum of a photon having energy ( is given as = =
So, …………….(1)
Where, Wavelength of the electromagnetic radiation
h = Planck's constant
c = speed of light
De Broglie wavelength of the photon is given as
But momentum, , where = mass of the photon, = velocity of the photon
Hence ……….(2)
Hence, it can be inferred from equation (1) and (2) that wavelength of the electromagnetic radiation is equal to the De Broglie wavelength of the photon.
New answer posted
11 months agoContributor-Level 10
11.17 De Broglie wavelength of the neutron, = 1.40 m
Mass of neutron, m = 1.66 kg
Planck's constant, h = 6.626 Js
Kinetic energy, = ………….(1)
De Broglie wavelength and velocity (v) are related as
…………….(2)
Combining equation (1) and (2), we get
= = = = 6.75 J = eV = 42.17
Temperature of the neutron, T = 300 K
Average kinetic energy of the neutron, = ,
where k = Boltzmann constant = 1.38 kg
= = J
The relationship of De Broglie wa
New answer posted
11 months agoContributor-Level 10
11.16 Wavelength of electron, = Wavelength of proton, = 1.0 nm = 1 m
Planck's constant, h = 6.626 Js
From De Broglie wavelength relation, where p = momentum
= = = 6.626 kg.m/s. Since , their momentum will be also equal.
The energy of photon is given by the relation: =
where c = speed of light = 3 m/s
= Js = eV = 1242.38 eV = 1.242 keV
Kinetic energy of electron, having momentum p is given by the relation
= where m = mass of electron = 9.1 kg.
Hence, &nbs
New answer posted
11 months agoContributor-Level 10
11.15 Mass of the bullet, m = 0.04 kg
Speed of the bullet, v = 1.0 km/s = 1000 m/s
Planck's constant, h = 6.626 Js
De Broglie wavelength of the bullet is given by the relation:
= = 1.65 m
Mass of the ball, m = 0.06 kg
Speed of the ball, v = 1.0 m/s
Planck's constant, h = 6.626 Js
De Broglie wavelength of the bullet is given by the relation:
= = 1.10 m
Mass of the dust particle, m = 1.0 kg
Speed of the dust particle, v = 2.2 m/s
Planck's constant, h = 6.626 Js
De Broglie wavelength of the bullet is given by the relation:
= = 3.01
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers