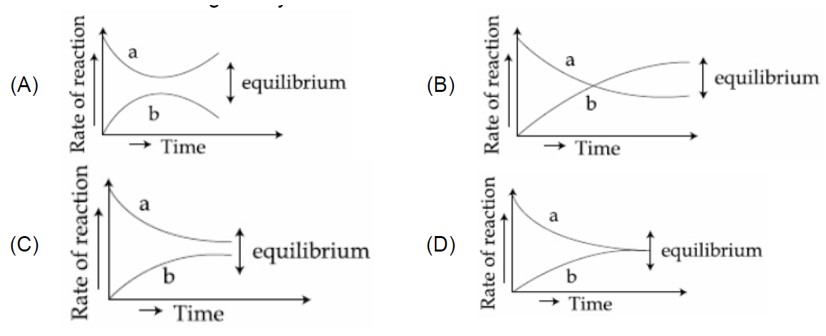
Chemical Equilibrium
Get insights from 78 questions on Chemical Equilibrium, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemical Equilibrium
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
In endothermic reaction formation of reactants is favoured upon decrease in temperature. Addition of inert gas at constant volume and temperature has no effect on equilibrium.
New answer posted
8 months agoContributor-Level 10
(x/m) = k (P)¹/?
log (x/m) = log k + (1/n) log P
Slope = 1/n = 2 So n = ½
Intercept ⇒ log k = 0.477 So k = Antilog (0.477) = 3
So (x/m) = k (P)¹/? = 3 [0.04]² = 48 * 10?
New answer posted
8 months agoNew answer posted
8 months agoContributor-Level 10
(b) It is harmful for trees and plants
(c) It causes breathing problem in human being and animals
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers