Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans: CH3CH2CH3 < CH3CH2NH2 < CH3CH2OH
As oxygen is more electronegative than nitrogen therefore, the O-H bond is more polar than the N-H bond. So ethanol has more dipole moments than ethylamine. Propane is non- polar in nature hence, had the least among all.
New answer posted
9 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(a, c, d)
(a) Cationic detergents are quaternary ammonium salts of amines with acetates, chlorides or bromides as anions. These detergents have germicidal properties.
(b) Bacteria cannot degrade the detergents containing highly branched chains, therefore, in most of the detergents used these days, the branching is kept to a minimum so that the detergents become easily biodegradable.
(c) Some synthetic detergents can give foam even in ice cold water.
(d) Synthetic detergents are cleansing agents which have all the properties of soaps, but which actually do not contain any
New answer posted
9 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans: CS2 is a non-polar solvent which decreases the activating effect of? NH2. As a result, mono substitution occurs only at o- and p- positions giving a mixture of 2-bromoaniline (minor) and 4-bromoaniline (major) as products.
New answer posted
9 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
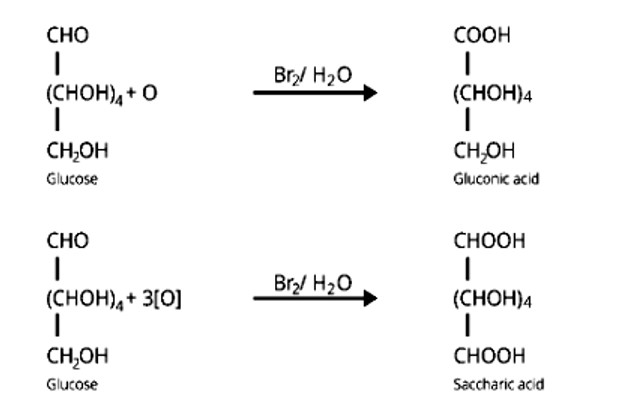
Monosaccharides contain carbonyl groups. As a result, they fall under the aldose or ketose categories. When an aldehyde group is present, the monosaccharides are known as aldose, and when a ketone group is present, the monosaccharides are known as ketose. Fructose is classified as ketohexose because it has six carbons and a keto group in its chemical formula (C6H12O6)
New answer posted
9 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans: The azo products have an extended conjugate system with both aromatic rings linked by the –N=N- bond. These compounds are frequently colored and used as dyes. Benzene diazonium chloride reacts with phenol to form p-hydroxy azobenzene by coupling the phenol molecule in its para position with the diazonium salt. This is referred to as a coupling reaction.
New answer posted
9 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans: Amides are the byproducts of the acylation reaction. The reaction is carried out in the presence of a stronger base than the amine, such as pyridine, which removes the formed HCl and shifts the equilibrium to the right.
New answer posted
9 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans: As the electronegativity of oxygen is more than the electronegativity of a nitrogen atom, the O−H bond is more polar than the N−H bond, therefore MeOH is stronger acid than MeNH2 or MeNH2 is stronger base than MeOH.
New answer posted
9 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
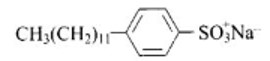
(a, d) Sodium salts of sulphonated long chain alcohol and sodium salts of sulphonated long chain hydrocarbons are anionic detergents e.g., Sodium laurylsulphate CH3 (CH2)10CH2OSO3– Na+ and sodium dodecylbenzene sulphonate.
New answer posted
9 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans: As −NH2 is a strong activating group, the aniline will readily undergo electrophilic substitution reaction, and it is difficult to cease reaction at the mono substitution stage.
Therefore, the activating group −NH2 is protected by an acetylation process.
The acetylated complex formed utilizes the lone pair of nitrogen and are less available for donation, this helps to carry out the nitration reaction easily.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers