Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Physical or chemical changes damage hydrogen bonding and other attractive factors. In addition, globules uncoil and the helix uncoils, resulting in a thread-like molecule. As a result, the biological activity of protein secondary and tertiary structures is lost entirely or partially. Protein denaturation is the term for this.
New answer posted
9 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
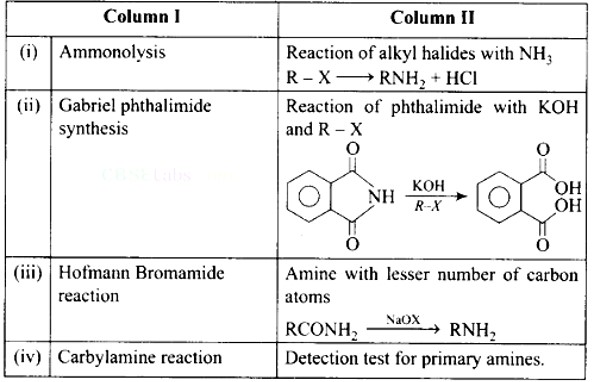
Ans: (i)- (d), (ii)- (c), (iii)- (a), (iv)- (b)
New answer posted
9 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Since both acidic (−COOH) and basic (−NH2) groups present in amino acids, it act like salts rather than simple amines or carboxylic acids. In Ans, a −COOH a group can release a proton and an amine group can take a proton, resulting in the formation of a dipolar ion known as zwitterion.
New answer posted
9 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans:
New answer posted
9 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Sucrose acts as a dextrorotatory molecule. It produces a mixture of glucose and fructose when hydrolyzed, with particular rotations of +52.5? and −92.4? The net resultant mixture is laevorotatory because fructose laevorotation is greater than glucose dextrorotation. The sign of rotation shifts from dextro to laevo as a result of sucrose hydrolysis, and the resulting compound is known as invert sugar.
New answer posted
9 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans:
New answer posted
9 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Excess vitamin C is easily removed in the urine since it is water soluble. As a result, it can't be stored in the body and must be ingested frequently.
New answer posted
9 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans:
New answer posted
9 months agoContributor-Level 10
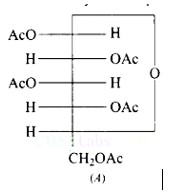
This is a Short Answer Type Questions as classified in NCERT Exemplar
The given structure A is glucose pentaacetate, which lacks a free hydroxyl? OH or carbon -CHO group. As a result, it cannot be transferred to the open chain form to make a free group, and therefore the oxime is not produced.
New answer posted
9 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans:
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers