Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
Due to their high electronegativity, small atomic size, greater ability to form multiple bonds, and absence of d-orbitals, the first elements of each group behave differently.
New answer posted
9 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Standard hydrogen electrode (SHE) is used as a reference electrode and its electrode potential is assumed to be zero. The electrode potential of other electrodes is measured concerning standard hydrogen electrodes.
New answer posted
9 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
The reaction's activation energy is relatively high at room temperature and not readily available, the reaction is highly viable. H2 (g) and O2 (g)bond break and clash, allowing particles to pass across the energy barrier.
2H2 + O2→2H2O
Because of the large activation energy, the water molecule does not form in this equation.
New answer posted
9 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Conductivity due to the total number of ions present in the solution is termed as specific conductivity. An increase in the number of ions per unit volume increases. In addition to water, the number of ions per unit volume decreases therefore conductivity decreases.
New answer posted
9 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
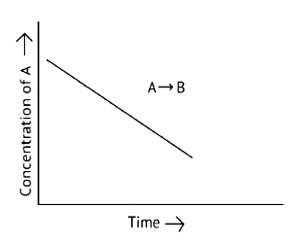
A. The reaction has a zero order. The concentration of the reactants does not change with time in a zero order reaction, and the rate of concentration remains constant
B. Slope of the curve is - k
C. The rate constant's units are molL 1s - 1
New answer posted
9 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: pH of the substance depends on the concentration of hydronium ions present. Here the concentration of hydronium ions remains constant so pH remains constant.
The electrolysis reaction is as follows;
At anode: 2H2O (l)→O2 (g)+ 4H+ + 4e−
At cathode:4H+ + 4e− →2H2
New answer posted
9 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Stronger electrolytes undergo cent percent dissociation. Electrolyte B will be the stronger electrolyte since the concentration of ions remains the same upon dilution.
New answer posted
9 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The number of ions present in the cell decides the life-time of any cell. In mercury cells, ions are not involved so the mercury cell has a constant cell potential throughout its useful life.
New answer posted
9 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Electrolysis of brine solution is as follows
NaCl (aq)→Na+ (aq)+Cl− (aq)
At the cathode, the reaction goes this way,
H2O (l) + e−→12H2 (g)+OH− (aq)
At the anode, the reaction goes this way,
Cl− (aq)→12Cl2 (g) + e−
The pH of the solution will increase as sodium hydroxide is being formed.
New question posted
9 months agoTaking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers