Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
53. (ii) Both assertion and reason are true but reason is not the correct explanation of assertion.
Explanation: Froth Flotation method is used to separate the hydrophobic materials from hydrophilic materials. In this method a mixture of palm oil, water and detergent is taken in a tank along with powdered sulfide ore. Compressed air is then passed through the pipe of the rotating agitator to create froth. The sulfide ore is then wetted by the palm oil mixture and it rises with the froth and the impurities or gauge settles at the bottom of the tank. The froth containing the sulfide is then cleaned and dried. Cresols or Aniline
New answer posted
10 months agoContributor-Level 10
34. (B) 3d5
Magnetic moment of an electron/dipole moment is caused by its intrinsic properties of spin and electric charge. It depends upon the number of unpaired electrons in its valence shell. The more the number of unpaired electrons, the greater will be the value of magnetic moment.
3d7 = 3 unpaired electrons
3d5 = 5 unpaired electrons
3d8 = 2 unpaired electrons
3d2 = 2 unpaired electrons
Out of all, 3d5 has five unpaired electrons that is maximum and hence it has the highest magnetic moment.
New answer posted
10 months agoContributor-Level 10
52. (i) Both assertion and reason are true and reason is the correct explanation of assertion.
Explanation: Van Arkel method is generally used to obtain pure forms of Zirconium (Zr) and Titanium (Ti).
The metal iodide is heated at 1800 K and is decomposed on a tungsten filament. The pure metal is dropped on the tungsten filament. This proves that ZrI4 is volatile and decomposes at 1800 K.
New answer posted
10 months agoContributor-Level 10
51. (i) Both assertion and reason are true and reason is the correct explanation of assertion.
Explanation: In the Mond process Nickel is reacted with carbon monoxide reversibly to give Nickel carbonyl, Ni (CO)4. Nickel carbonyl is a volatile compound. It decomposes to nickel and carbon monoxide at 460 K.
New answer posted
10 months agoContributor-Level 10
33. (A) Mn2O7
2KMnO4 + 2H2SO4 (conc.) → Mn2O7 + 2KHSO4 + H2O
Manganese heptoxide is an acid anhydride of permanganic acid ( HMnO4). It is a dangerous oxidiser, volatile and highly reactive liquid.
New answer posted
10 months agoContributor-Level 10
50. Option (i) A (2) B (3) C (4) D (1)
Explanation: A solidified copper has a blistered appearance due to the evolution of SO2 Hence it is called blistered copper.
Hence, option (A) from column I is matched with option (2) from column II. Iron is extracted from a blast furnace.
Hence, option (B) from column I is matched with option (3) from column II. The iron ore is heated in the reverberatory furnace after mixing with silica. In the furnace, iron oxide slags of iron and copper are produced in the form of copper matte.
Hence, option (C) from column I is matched with option (4) from column II. The hall-Heroult process is used for the extra
New answer posted
10 months agoContributor-Level 10
32. (B) CuF2
(A) Ag2SO4 (Ag+ )→ 5d106s0
(B) CuF2 (Cu2+ )→ 3d94s0
(C) ZnF2 (Zn2+)→ 3d104s0
(D) Cu2Cl2 (Cu+)→ 3d106s0
Unpaired electrons present in any compound impart colour to the salt of transition metal. Only CuF2 has unpaired electrons in its 3d orbital that's why it is white coloured in its solid-state while rest of the salts are colourless.
New answer posted
10 months agoContributor-Level 10
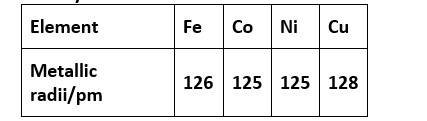
31. (D) Cu
Density is mass by volume. As we move from left to right for a long period, the atomic radii decrease. Hence, volume decreases. Also, an increase in atomic masses is observed.
So overall the density increases out of the above option from iron to copper, copper will have the highest density.
New answer posted
10 months agoContributor-Level 10
49. Option (i) A (3) B (4) C (2) D (1)
Sapphire is a gemstone containing Co.
Hence, option (A) from column I is matched with option (3) from column II. The Sphalerite single is ZnS.
Hence, option (B) from column I is matched with option (4) from column II. NaCN is also used as a depressant.
Hence, option (C) from column I is matched with option (2) from column II. Al2O3 is also called corundum.
Hence, option (D) from column I is matched with option (1) from column II.
New answer posted
10 months agoContributor-Level 10
30. (A) Cu (II) is more stable .
Electronic configuration of Cuis [Ar] 3d10 4s1
Cu (I) - [Ar] 3d10 4s0
Cu (II)- [Ar] 3d9 4s0
Despite the fact that Cu (I) has fully filled 3d-orbital but Cu (II) is more stable than Cu (I) due to the greater effective nuclear charge of Cu (II) as nucleus has to hold 17 electrons rather than 18 electrons like in Cu (I).
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers