Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
16.26
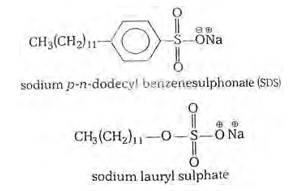
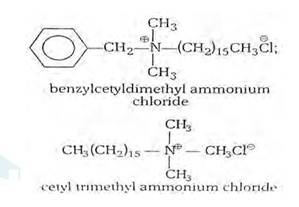
(i) Cationic detergents- Cationic detergents are quaternary ammonium salts [chlorides, bromides, acetates, etc.] having long chain alkyl groups. The cationic detergents are more expensive than anionic detergents and hence they find only limited use. However, they possess germicidal properties and are used quite extensively as germicides. The examples of cationic detergents are as follows:
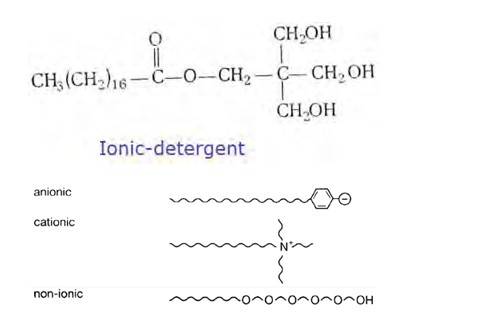
(ii) Anionic detergents- A detergent is said to be anionic when the large part of its molecule is an anion and is involved in the cleansing action. The anionic detergents are also effective in slightly acidic In slightly acidic solutions,
New answer posted
11 months agoContributor-Level 10
Let the total mass of the solution be 100 g and the mass of benzene be 30 g.
∴ Mass of carbon tetrachloride = (100 - 30) g = 70 g
Molar mass of benzene (C6H6) = (6 * 12 + 6 * 1) g mol -1
= 78 g mol -1
∴ Number of moles of C6H6 =30/78 mol
= 0.3846 mol
Molar mass of carbon tetrachloride (CCl4) = 1 * 12 + 4 * 35.5
= 154 g mol -1
∴ Number of moles of CCl4 = 70/154 mol
= 0.4545 mol
Thus, the mole fraction of C6H6 is given as:
Number of moles of C6H6 / Number of moles of C6H6 + Number of moles of CCl4
= 0.3846 / (0.3846 + 0.4545)
New answer posted
11 months agoContributor-Level 10
Mass of Solution = Mass of Benzene + Mass of Carbon Tetrachloride
= 22 g + 122 g = 144 g
Mass percentage of Benzene = Mass of Benzene / Mass of Solution X 100 = 22/144 X 100 = 15.28%
Mass percentage of CCl4 = Mass of CCl4 / Mass of Solution X 100 = 122/144 X 100 = 84.72%
New answer posted
11 months agoContributor-Level 10
16.25
Synthetic detergents are superior cleansing agents as compared to soaps. This is due to the following reasons.
1. Detergents are soluble even in hard water. This is because of calcium and magnesium ions present in hard water form soluble salts with detergents. Hence, detergents can be used both in soft as well as in hard water. On the other hand, soaps form insoluble salts with calcium and magnesium ions and cannot be used in hard water.
2. The aqueous solutions of detergents are usually neutral. Therefore, they do not damage delicate fabrics and can be used for washing almost all types of On the other hand, aqueous solu
New answer posted
11 months agoContributor-Level 10
Given-
Mass of K2SO4, w = 25 mg = 25 X 10-3 g,
Molar mass of K2SO4 = (39*2) + (32*1) + (16*4) = 174 g mol-1
Volume V = 2 liter
T = 250C + 273 = 298 K (add 273 to convert in Kelvin)
The reaction of dissociation of K2SO4 is written as,
K2SO4 → 2K + + SO42-
Number if ions produced = 2 + 1 = 3, hence vant Hoff's factor, I = 3
Here, we use vant Hoff's equation for dilute solutions, given as,
πV = inRT
where, n is the number of moles of solute, R is solution constant which is equal to the gas constant (0.082) and T is the absolute temperature (298 K).


Hence, the osmotic pressure of a solution is 5.27x10-3atm
New answer posted
11 months agoContributor-Level 10
16.24
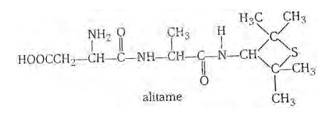
Alitame is a high potency artificial sweetener and it is not possible to control the sweetness imparted to food by alitame. Alitame is an artificial sweetener that is 2,000 times as sweet as sugar. This sweetness is very high as compared to the natural sugar and use of such sweetener is very critical while preparing sweet dishes. The structure of alitame is as follows:
New answer posted
11 months agoContributor-Level 10
Given-
Vant Hoff's factor, I = 2.47
osmotic pressure, π = 0.75 atm
Volume of solution = 2.5L.
To determine the amount of CaCl2, we use vant Hoff's equation for dilute solutions, given as,
πV = inRT
where, n is the number of moles of solute, R is solution constant which is equal to the gas constant and T is the absolute temperature.

Hence, the amount of CaCl2 dissolved is 3.425g
New answer posted
11 months agoContributor-Level 10
Given-
KH for O2 = 3.30 * 107 mm Hg,
KH for N2 = 6.51 * 107 mm Hg
Percentage of oxygen (O2) = 20 %
Percentage of nitrogen (N2) = 79%
Total pressure = 10 atm
Using Henry's law,

where, p is the partial pressure of gas in the solution and KH is Henry's constant.

Thus, the mole fraction of oxygen in solution, xoxy = 4.61x10-5
and the mole fraction of nitrogen in solution, xnit is 9.22x10-5
New answer posted
11 months agoContributor-Level 10
16.23
Artificial sweetening agents like Saccharin, Alitame, Sucrolose can be used in the preparation of sweets for a diabetic patient as they do not add any calories to the body.
Diabetic people are advised to consume low-calorie diet [fewer carbohydrates & more proteinaceous and fiber rich]. The refined sugar like sucrose adds calorie to the diet but complex sugar and starches do not add to the calorie intake of a person and at the same time impart a sweet taste to diet. Artificial sweeteners are generally either complex sugar or protein in nature
New answer posted
11 months agoTaking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers