Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a matching answer type question as classified in NCERT Exemplar
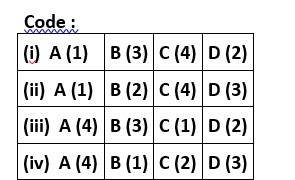
Correct option is (i)
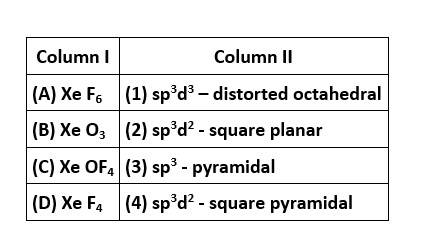
In XeF6, the central Xe atom undergoes sp3d3 hybridisation and the molecular geometry is distorted octahedral with 1 lone pair and 6 bond pairs of electrons.
In XeO3, the central Xe atom undergoes sp3 hybridisation and the molecular geometry is pyramidal with 1 lone pair and 3 bonding domains of electrons.
In XeOF4, the central Xe atom undergoes sp3d2 hybridization and the molecular geometry is square pyramidal with 1 lone pair and 5 bond pairs of electrons.
In XeF4, the central Xe atom undergoes&nb
New answer posted
10 months agoContributor-Level 10
This is a Objective Type Question as classified in NCERT Exemplar
Sol:
New answer posted
10 months agoContributor-Level 10
This is a Objective Type Question as classified in NCERT Exemplar
Sol:
New answer posted
10 months agoContributor-Level 10
This is a assertion and reason answer type question as classified in NCERT Exemplar
Correct option is (i)Here, the S atom is surrounded by 6F atoms, so it's complected for H2O to attack SF6, but in case of SF4, it's surrounded by 4F atoms, so it's to attack H2O.
New question posted
10 months agoNew answer posted
10 months agoContributor-Level 10
This is a assertion and reason answer type question as classified in NCERT Exemplar
Correct option is (i)
When NaCl reacts with concentrated H2SO4, it produces colourless fumes with a pungent smell, but when MnO2 is added, the fumes turn greenish yellow. MnO2 oxidizes HCl to chlorine gas, which then turns greenish yellow.
New answer posted
10 months agoContributor-Level 10
This is a Objective Type Question as classified in NCERT Exemplar
Sol:
New answer posted
10 months agoContributor-Level 10
This is a assertion and reason answer type question as classified in NCERT Exemplar
Correct option is (i)
The rhombic and the monoclinic forms of sulphur exist as S8 . Oxygen forms a p\pi - p\pi multiple bond due to its small size and short bond length, but Pπ- Pπ bonding is not possible because of sulphur's larger atomic size.
New answer posted
10 months agoContributor-Level 10
This is a Objective Type Question as classified in NCERT Exemplar
Sol:
New answer posted
10 months agoContributor-Level 10
This is a assertion and reason answer type question as classified in NCERT Exemplar
Correct option is (ii)
Because H2SO4 is a stronger oxidising agent and HI is a stronger reducing agent, H2SO4 oxidizes and is reduced to SO2.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers