Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Correct option is (iv)
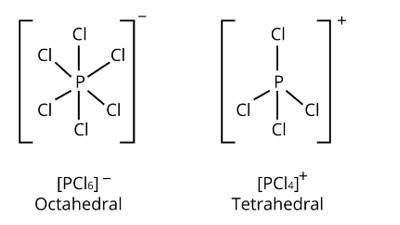
The ionic bonding promotes the crystalline structure of PCl5 in the solid state by attempting to exist as oppositely charged ions like [PCl4]+ and [PCl6]− . Also, [PCl4]+ and [PCl6]− are tetrahedral and octahedral, respectively. These structures fit together well, giving the solid structure extra stability.
New answer posted
10 months agoContributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Sol:
New answer posted
10 months agoContributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Sol:
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Correct option is (iii)
Bartlett noticed that Xenon's first ionisation potential is nearly identical to that of oxygen and used Born-Haber calculations to predict the presence of a stable compound: Xe+ Pt F6-
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Correct option is (iv)
MnO2 ( Black )+4HCl→MnCl2+2H2O+Cl2
Greenish yellow color
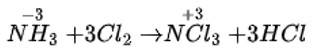
New answer posted
10 months agoContributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Sol:
New answer posted
10 months agoUsing the definition, prove that the function is invertible if and only if is both one-one and onto.
Contributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Sol:
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Correct option is (iii)
C+2H2SO4→CO2+2SO2+2H2O
Conc. H2SO4 oxidises C to produce two gaseous products.
New answer posted
10 months agoContributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Sol:
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
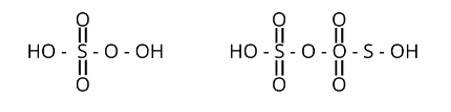
Correct option is (i)
Peroxy linkage refers to the presence of a bond between oxygen and oxygen (O-O) in a molecule.
Only H2S2O6 and H2S2O7 of the sulphur oxoacids above have a peroxy linkage.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers
