Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Correct option is (ii) and (iv)
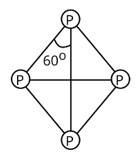
Tetrahedral geometry characterises the structure of a white phosphorus molecule.
Phosphorus has a valency of 5, and there are four phosphorus atoms in total. As a result, there will be a total of 4*5=20 valence electrons.
As a result, each P atom will have one lone pair of electrons, and each covalent bond (two electrons) will be established.
In a molecule of white phosphorus, there will be four lone pairs of electrons and six P-P single bonds in total.
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Correct option is (ii), (iii)
MI > MBr > MCl > MF is an iconic character of metal halide
F2 > Cl2 > Br2 > I2 is a bond dissociation enthalpy
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
Sol:
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
Sol:
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Correct option is (i) and (iii)
6NaOH+3Cl2 → 5NaCl+NaClO3+3H2O
Chlorine gas oxidation number ranges from 0 to –1 to 0 to +5.
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
Sol:
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
Sol:
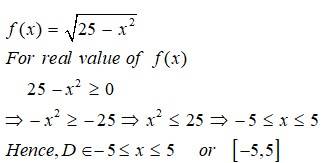
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Correct option is (ii)
BrO2- (35+2*8+1=52) and BrF2+ (35+2*9−1=52)
52 electrons are present in both
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
Sol: R = { (a, a), (b, c), (a, b)}.
To make R as reflexive we must add (b, b) and (c, c) to R. Also, to make R as transitive we must add
(a, c) to R.
So, minimum number of ordered pair is to be added are (b, b), (c, c), (a, c).
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Correct option is (iii)
The ability of a substance to be decreased is known as its reduction potential. As the substance's reduction potential rises, so does its power to reduce. It signifies the chemical's oxidising power (the ability of the material to cause other substances to lose electrons, which is known as oxidising power) grows.
As a result, the oxidising power is listed in decreasing order BrO4- > IO4- > ClO4-
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers


