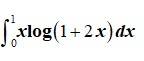Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Sol:
New answer posted
11 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar

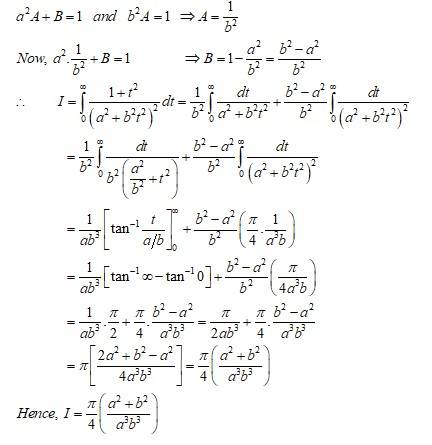
New answer posted
11 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar


New answer posted
11 months agoContributor-Level 10
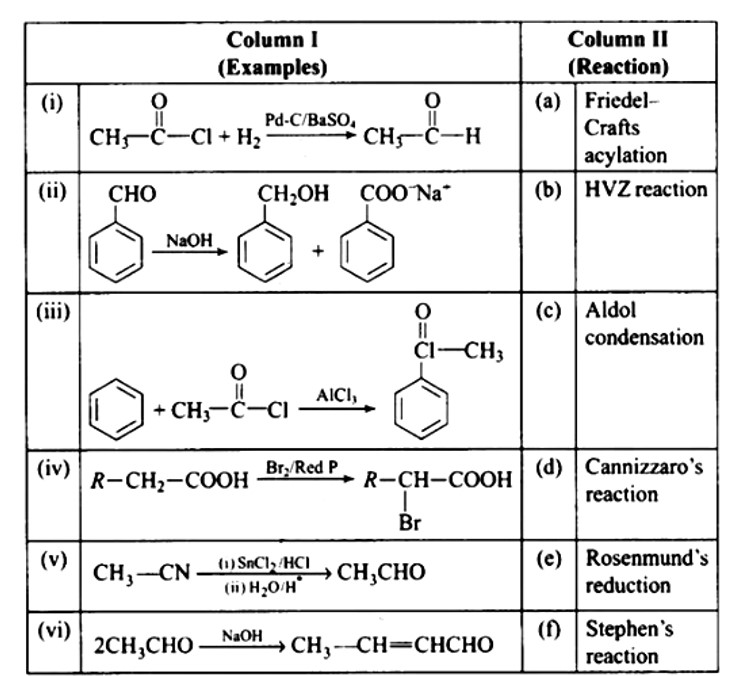
This is a Matching Type Questions as classified in NCERT Exemplar
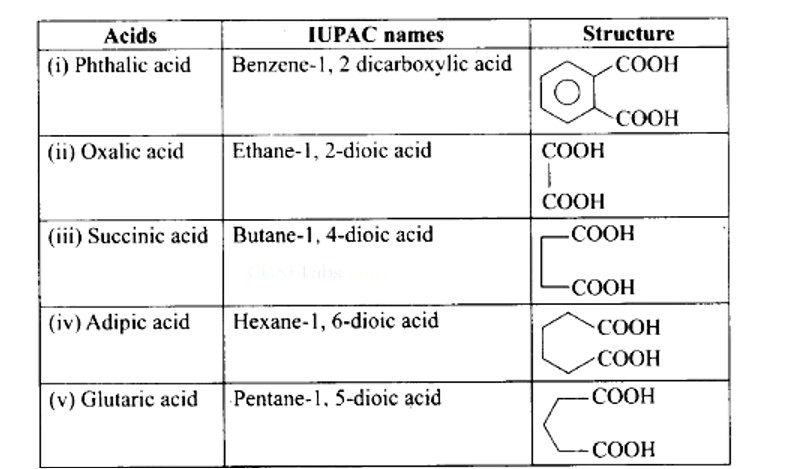
Ans: (i) — (e); (ii) — (d); (iii) — (a); (iv) — (b); (v) — (f); (vi) — (c)
New answer posted
11 months agoContributor-Level 10
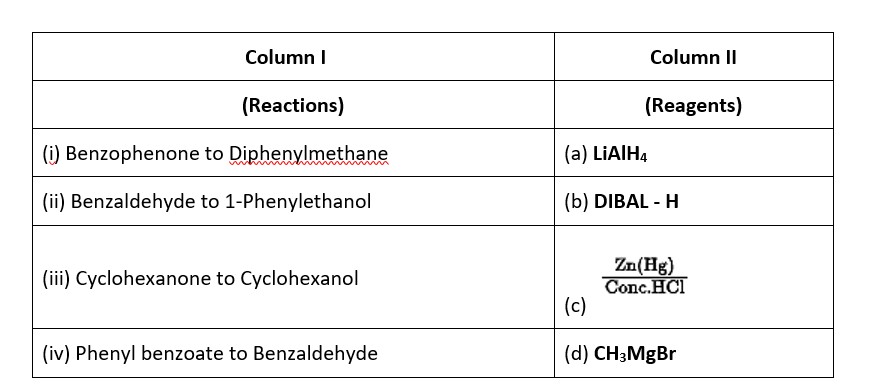
This is a Matching Type Questions as classified in NCERT Exemplar
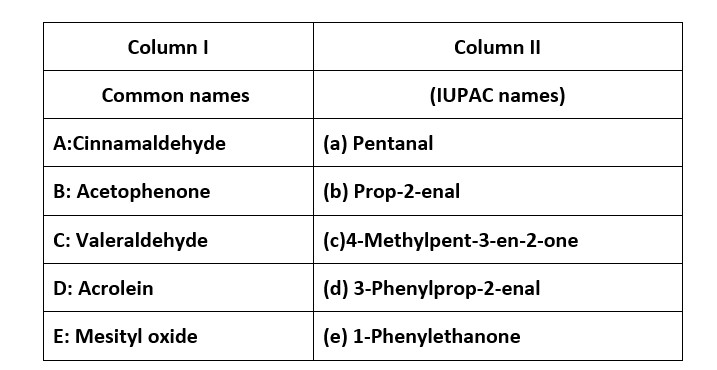
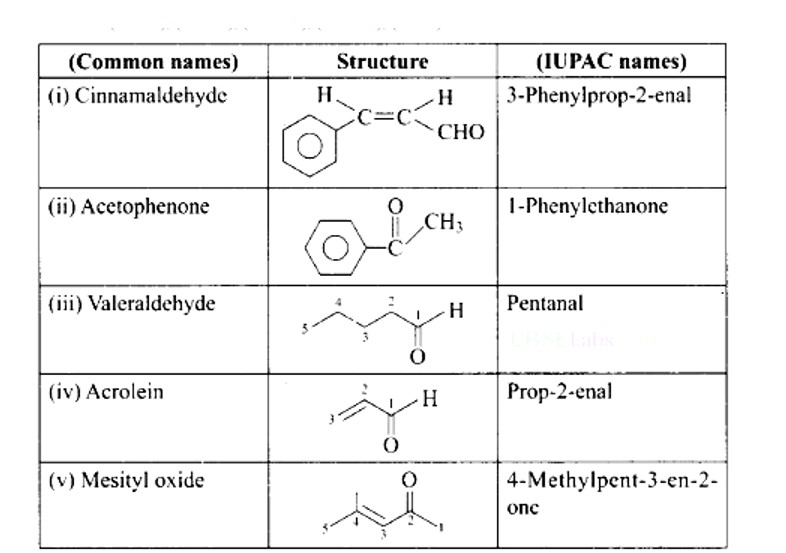
Ans: (i) → (c); (ii) → (d); (iii) → (a); (iv) → (b).
New answer posted
11 months agoContributor-Level 10
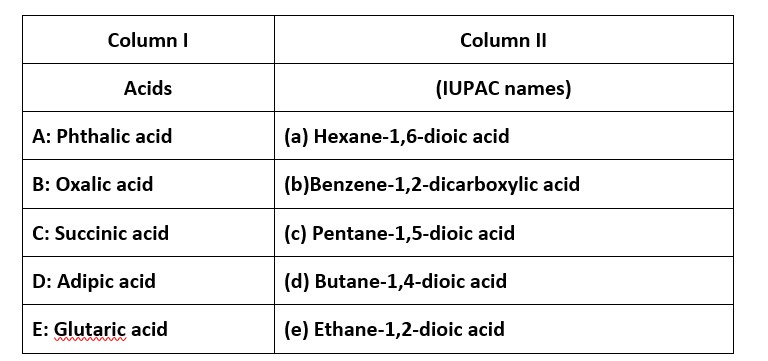
This is a Matching Type Questions as classified in NCERT Exemplar
Ans: (i) → (b); (ii) → (e); (iii) → (d); (iv) → (a); (v) → (c)
New answer posted
11 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans: (i) → (d); (ii) → (e); (iii) → (a); (iv) → (b); (v) → (c)
New answer posted
11 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: D
The silver mirror test can be used to determine Tollen's. [Ag (NH3)2] + OH - . Only aldehydes, not ketones, react with Tollen's reagent to create silver.
A silver mirror test is not given with this affirmative test.
The carbonyl group is present in both aldehyde and ketone.
New answer posted
11 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: C
There is no alpha hydrogen atom in the Cannizaro process. Formaldehyde and aromatic aldehydes do not contain alpha hydrogen. It proceeds through the Cannizaro reaction. Formaldehyde is the most reactive of all aldehydes.
New answer posted
11 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: D
Because of the presence of electron withdrawing carbonyl groups, the alpha hydrogen atom in carbonyl compounds is acidic. In nature, hydrogen is very acidic.
Because the cation is released in the form of H+ , the anion created after the loss of the -hydrogen atom is resonance stabilised.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers