Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
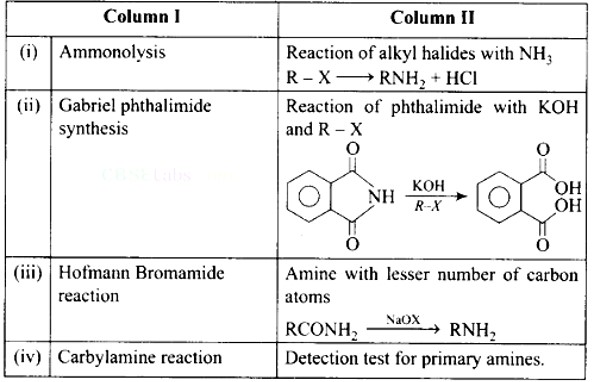
This is a Matching Type Questions as classified in NCERT Exemplar
Ans: (i)- (d), (ii)- (c), (iii)- (a), (iv)- (b)
New answer posted
9 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans:
New answer posted
9 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans:
New answer posted
9 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
When an egg is cooked, the soluble globular protein albumin transforms into an insoluble fibrous protein. Bioactivity is lost throughout the denaturation process.
(ii) Albumin protein's secondary and tertiary structures are destroyed, but the primary structure (which contains the -amino acid sequence) is unaffected.
Although egg white is a fluid Ans, when it is cooked, the proteins get denatured and solidify as a result of coagulation.
New answer posted
9 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (d)
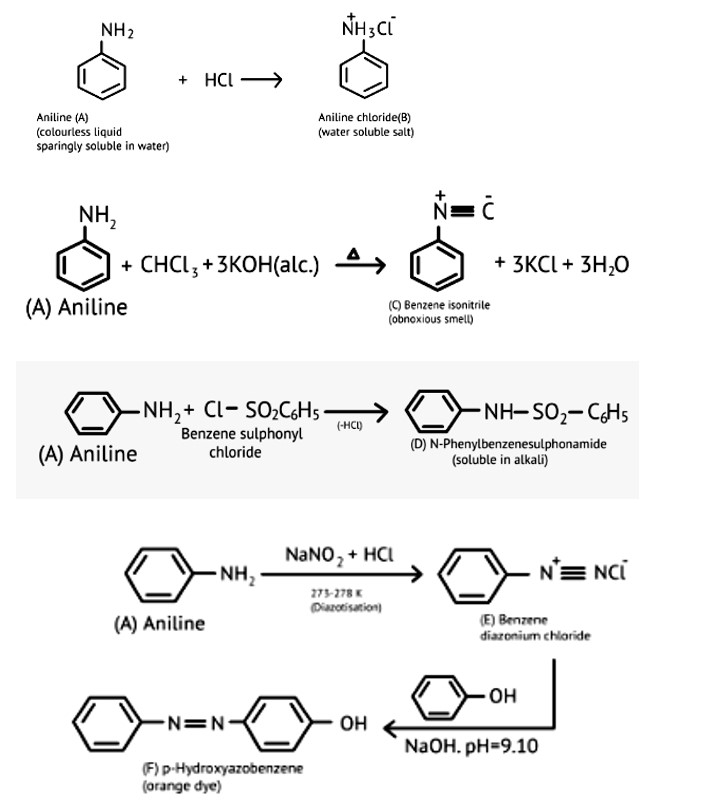
Acetanilide is less basic than aniline because electron density of nitrogen is lowered by acetyl group.
New answer posted
9 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (a)
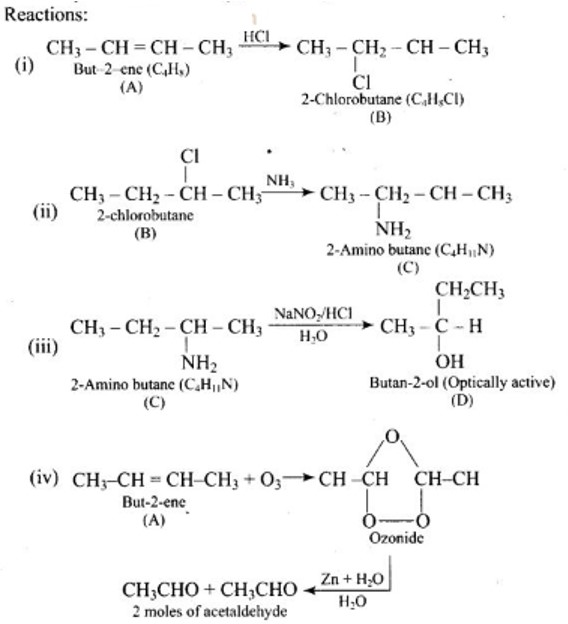
Aromatic primary amines cannot be prepared using Gabriel phthalimide synthesis as aryl halides do not undergo nucleophilic substitution with anion formed by phthalimide. Both the statement's assertion and reason are incorrect, hence the correct answer is a.
New answer posted
9 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
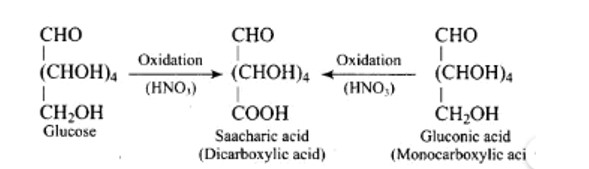
Glucose with acetic anhydride in the presence of pyridine or a few drops of concentrated H2SO4, leads to productions of penta-acetyl derivatives which shows that there are five hydroxyl groups. One hydroxyl group is a primary (1? ) alcoholic, whereas the other four (C2, C3, C4, and C5) hydroxyl or −OH groups are all labelled as secondary (2? ) alcoholics.
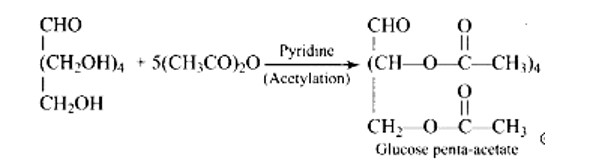
When glucose (or gluconic acid) is oxidized in the presence of HNO3, it produces saccharic acid (a dicarboxylic acid), showing that the primary (1? ) alcoholic group is oxidized to −COOH, but only under extreme of
New answer posted
9 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (d)
Fe+2HCl→FeCl2+2 [H] FeCl2+H2O (g)→FeO+2HCl
The nascent hydrogen formed act as reducing agent for the reduction of nitro compounds.
New answer posted
9 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (b)
N, N-die ethyl benzene sulphonamide, there is no acidic hydrogen present on the N-atom which can make it soluble. Therefore, it is insoluble in alkali.
New answer posted
9 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Sugars are mainly classified into two configurations: D− configuration and L− configuration, each of which have individual distinct structures. These configurations are expressed in terms of the standard glyceraldehyde which are basically of two types:
D− (+)− Glyceraldehyde and L− (−)− Glyceraldehyde. The D− configuration has −OH attached to the carbon near to −CH2OH on the right, whereas the L− configuration has −OH attached to the carbon adjacent to −CH2OH on the left. The sugars are designated D− and L− depending on whether the molecul
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers