Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
This is a Fill in the blanks Type Question as classified in NCERT Exemplar
Ans: Correct option A
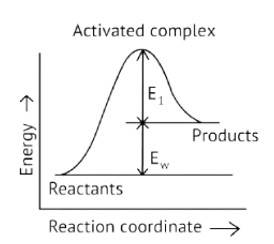
Activation Energy → The amount of energy required to overcome the obstacle and generate a product
The activation energy of a forward reaction can be seen in the [Eaf = E1 + E2] (This is an endothermic reaction.
Therefore, [Eaf > Eab]
The energy of the product is high, while the energy of the reactant is low.
The lower the energy, the more stable and positive the situation becomes.
Δ? = posistive
New answer posted
9 months agoContributor-Level 10
The NCERT Exemplar includes analytical, application-based, and reasoning-type questions. It goes beyond the NCERT textbooks and helps students apply theories like VSEPR, hybridization, and MOT. It helps students to prepare for various entrance exams like NEET and JEE by strengthening their conceptual clarity.
New answer posted
9 months agoContributor-Level 10
Hybridization predicts the geometry and bond angles in complex molecules and it helps in the understanding of how four equivalent bonds in methane (CH? ) can be formed from atoms like carbon. For observed molecular geometries, it explains the formation of the equivalent orbitals (like sp³, sp², sp).
New answer posted
9 months agoContributor-Level 10
In molecular shape prediction, VSEPR (Valence Shell Electron Pair Repulsion) theory is important because based on the repulsions between the electron pairs around the central atom, it helps in predicting the three-dimensional shape of molecules. The 3D shape influences the molecule's chemical and physical properties including reactivity, polarity, and intermolecular forces.
New answer posted
9 months agoContributor-Level 10
This is a assertion and reason answer type question as classified in NCERT Exemplar
Because m-nitrochlorobenzene is not a stable molecule, the -NO2 group is a meta-directing group, and the reactions' products include nitro groups at the o- and p- positions.
Correct Answer: Option (iv)
New answer posted
9 months agoContributor-Level 10
This is a assertion and reason answer type question as classified in NCERT Exemplar
The SN2 mechanism is demonstrated by the hydrolysis of alkyl halides with inversion of configuration. This is a one-step method that does not need the production of carbocation.
Correct Answer: Option (iii)
New answer posted
9 months agoContributor-Level 10
This is a assertion and reason answer type question as classified in NCERT Exemplar
Due to resonance, a partial double bond character occurs in the bond between the C and Cl atoms in chlorobenzene, and we all know that changing a partial double bond character is more difficult than replacing a single bond as in the C - Cl bond in chloroethane.
Correct Answer: Option (i)
New answer posted
9 months agoContributor-Level 10
This is a assertion and reason answer type question as classified in NCERT Exemplar
Oxidising chemicals such as HIO3 oxidise HI to I2 because the presence of HI causes the aryl iodides to revert to arenes in their absence.
Correct Answer: Option (iii)
New answer posted
9 months agoContributor-Level 10
This is a assertion and reason answer type question as classified in NCERT Exemplar
Because halogen atoms are ortho and para directing, rather than ring deactivators, further electrophilic substitution occurs at ortho and para locations.
Correct Answer: Option (v)
New answer posted
9 months agoContributor-Level 10
This is a assertion and reason answer type question as classified in NCERT Exemplar
The nitro group on the ortho and para positions of haloarenes acts as an electron-withdrawing group, making it more reactive to nucleophilic substitution reactions due to the electron deficit.
Correct Answer: Option (i)
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers