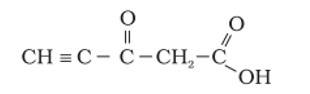Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (ii)
In a molecular structure if a central atom has the same hybridization as the central atom of another molecular species, then the two structures are known as isostructural species.
(i) In NF3 the central nitrogen atom is sp3 hybridized and in BF3 the central boron atom is sp2 . So, these two molecules are not isostructural pairs.
(ii) In BF4 the central boron atom is sp3 hybridized and in NH4 + the central nitrogen atom is also sp3 So, these two molecules are identified as isostructural pairs.
(iii) In BCl3 the central boron atom is sp2 hybridized and in
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The average bond enthalpy is defined as the ratio of total bond dissociation enthalpy to the number of bonds broken in the structure.
The identical O- H bonds in water molecule does not have the same bond enthalpies. According to the structure of a water molecule, there are two O- H bonds, but there is a change in the breaking of the first O- H bond than the second because of the different charge.
Hence in water molecule the average bond enthalpy will be:
The average O-H bond enthalpy = = 464 mol-1
In ethanol C2H5OH the bond enthalpy of O-H is different becaus
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
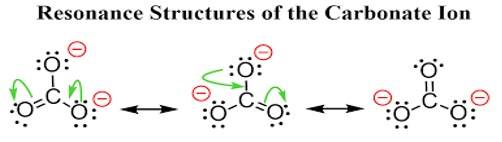
Ans: All the C-Obonds in carbonate ion (CO32-) are equal in length due to the equivalent resonance of all the three carbon-oxygen bonds gets a double bond character atleast once. This type of double bond character happened throughout 3C- O skeleton, and hence all the bonds acquired equal bond length.
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: In compound BCl3, Boron has sp2-hybridisation and the shape is Triangular Planar.
In methane CH4, Carbon has sp3 -hybridization and shape are Tetrahedral.
In carbon dioxide CO2, carbon has sp-hybridisation and shape is Linear.
In ammonia NH3, nitrogen has sp3-hybridisation and shape is Pyramidal.
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: (i)

(ii) 
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: (i) According to the electronic configuration, a total of 8 electrons must be present in the valence shell of an element. Element X has 4 valence electrons, so it will share the remaining 4 electrons for the formation of the bond, the molecular formula will be XH4 . The element Y has 5 valence shell electrons, so it will form 3 bonds and the formula will be YH3 . The element Z has 7 valence shell electrons, so it will form one bond with hydrogen and has the molecular formula H-Z .
(ii) Elements X, Y and Z having, 5 and 4 7 valence electrons respectively belongs
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: BeCl2 has a linear structure
HOCl is also non-linear in structure.
H2O has a V-shaped structure.
Cl2O has a V-shaped structure.
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The hybridization of Carbon 1 is sp, carbon 2 is sp, carbon 3 sp2, carbon 4 is sp3 and carbon 5 is sp2. The triple bond has 2 pie bonds and one sigma bond. Each double bond has one sigma and one pie bond. Every single bond is a sigma bond. Thus, the total number of sigma bonds is 11 and pie bonds are 4.
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The carbonate ion CO32- can be best represented by its resonating structures. The carbonate ion cannot be represented by a single Lewis structure because the three carbon oxygen bond lengths are the same. This cannot be shown by a single Lewis structure. For showing the similar lengths of all the carbon to oxygen bonds three hybrid structures are constructed which are in resonance with each other.
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
N—H, F—H, C—H and O—H
Ans: The ionic character in a molecular species is decided by the electronegativity difference between the two bonded atoms. Greater the electronegativity difference between two bonded pairs, the greater will be the ionic character. The electronegativity difference of the given species is
C - F = (2.5 - 2.1) = 0.4
N -H = (3.0 - 2.1) = 0.9
O - H = (3.5 - 2.1) = 1.4
F - H = (4.0 - 2.1) = 1.9
According to the above given difference in the electronegativities, the order of increasing ionic character is:
C – H< N - H
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers