Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
6. Interstitial compounds are formed when small atoms like H, C and N get trapped inside the crystal lattice of transition metals.
Characteristics:
They are hard and rigid in nature having high melting points.
Like pure metals only, they are conductors. They can gain chemical inertness.
New answer posted
10 months agoContributor-Level 10
21. Option (iii)
Copper ore is mixed with silica before heating in a reverberatory furnace. When heated in a reverberatory furnace, the iron oxide slags of iron silicate and copper are produced. This product is produced in the form of copper matte in the furnace. Presence of Cu2S and FeS is observed in copper matte. In these compounds, we observe the sulfides of Copper (I) Cu2+ and Iron (II) Fe2+.
New answer posted
10 months agoContributor-Level 10
5. (A) (i) Out of all the elements of the first transition series copper has the highest second ionisation enthalpy.
Electronic configuration of Copper is: 3d104s1
After the Loss of first electron from the 4s copper acquires 3d10 configuration which is stable. Therefore, removal of second electron from the field 3-D orbital is very difficult and requires high amount of energy.
(ii) Among the elements of first transition series zinc has the highest third ionisation enthalpy. Electronic configuration of zinc is: 3d104s2
After the loss of two electrons from 4s orbital, Z and +2 Ion acquires 3d10 fully filled configuration which is highly sta
New answer posted
10 months agoContributor-Level 10
4. (a) As per fajan's rule, smaller the size of ion, greater is its tendency to make covalent bonds as the positively charged cation strongly attract the negatively charged electron cloud.
On moving from La to Lu in the lanthanoid series, the atomic size decreases so the covalent character increases. Hence, La2O3 is ionic and Lu2O3 is covalent.
(b) The stability of Oxo salts is directly proportional to the size of atom, as we move from La to Lu, the size of the atom decreases and hence the stability of oxo salts also decreases.
(c) As we move along the lanthanide series, the atomic size decreases. As a result, the charge/size ratio incr
New answer posted
10 months agoContributor-Level 10
4. As per fajan's rule, smaller the size of ion, greater is its tendency to make covalent bonds as the positively charged cation strongly attract the negatively charged electron cloud.
On moving from La to Lu in the lanthanoid series, the atomic size decreases so the covalent character increases. Hence, La2O3 is ionic and Lu2O3 is covalent.
New answer posted
10 months agoContributor-Level 10
3. (A) = MnO2
(B) = K2MnO4
(C) = KMnO4
(D) = KIO3
2MnO2 + 4KOH + O2 → 2K2MnO4 + 2H2O
(A) (B)
3MnO42- + 4H+ → 2MnO4- + MnO2 + 2H2O
(C)
2MnO42- + 2H2O + KI→ 2MnO2 + 2SO4- + KIO3
(A)
New answer posted
10 months agoContributor-Level 10
2. (A) = FeCr2O4
(B) = Na2CrO4
(C) = Na2Cr2O7
(D) = K2Cr2O7
4FeCrO4 + 8NaCO3 + 7O2 → 8Na2CrO4 + 2Fe2O3 + 8CO2
(A) (B)
2NaCrO4 + 2H + → Na2Cr2O7 + 2Na + + H2O
Na2Cr2O7 + KCl→ K2Cr2O7 + 2NaCl
(C) &
New answer posted
10 months agoContributor-Level 10
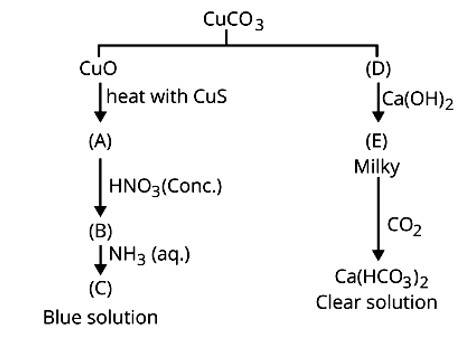
1. CuCO3 ![]() CuO + CO2
CuO + CO2
(D)
Ca (OH)2 + CO2 → CaCO3 + H2O
(E)
CaCO3 + CO2 + H2O → Ca (HCO3)2
clear sol.
CuO + CuS ![]() 3Cu + SO2
3Cu + SO2
(A)
Cu + 4HNO3 → Cu (NO3)2 + 2NO2 + 2H2O
&nb
New answer posted
10 months agoContributor-Level 10
77. Option (iii) Assertion is correct statement, but reason is wrong statement is the answer since intermediate conductivity in semiconductor is due to the small energy gap between valence band and conduction band and hence the assertion is correct, but the reason is wrong.
New answer posted
10 months agoContributor-Level 10
76. Option (ii) Assertion and reason both are correct statements, but reason is not correct explanation for assertion is the answer since in fcc structure the number of atoms present = 4 per unit cell which provides a maximum packing efficiency of 74%.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers