Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
6. In solids the constituent particles are found to possess fixed positions and can only oscillate about their mean positions and hence they are said to be incompressible and rigid.
New answer posted
10 months agoContributor-Level 10
5. The liquids and gases both are categorised as fluids as they show the ability to flow and also their molecules can move past one another freely.
New answer posted
10 months agoContributor-Level 10
4. Let the formula of sample be (Fe2+)x (Fe3+) yO
From the formula of the compound the equation can be formed as-
x+ y= 0.93 . (i)
Total positive charge on ferrous and ferric ions should balance the two units of negative charge on oxygen. Hence,
2x+ 3y= 2 . (ii)
⇒ x+ y=1. (iii)
On subtracting equation (i) from equation (iii) we have
y-y=1 - 0.93
12y=0.07
y= 0.14
On putting the value of y in equation (i)
we get, x+ 0.14 = 0.93
⇒x = 0.93 – 0.14
⇒x = 0.79
Fraction of Fe2+ ions present in the sample is = 0.81
Metal deficiency defect is found to be present in the sample
New answer posted
10 months agoContributor-Level 10
3. The conductivity of a semiconductor is very low and it can be increased by adding small impurities and this process is known as doping.
Doping can be done with an impurity which is electron rich or electron deficient-
n-type semiconductors- Si or Ge (group-14 elements) are doped with electron rich impurity (group-15 elements like P or As) and are known as n-type semiconductors. In them the conductivity is due to the extra electron or delocalized electron.
When intrinsic semiconductors like Si or Ge are doped with pentavalent elements like P or As, they occupy some of the lattice sites in silicon or germaniu
New answer posted
10 months agoContributor-Level 10
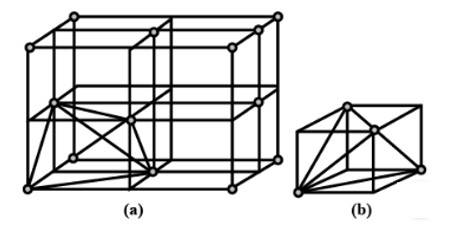
2. A ccp structure unit cell is divided into 8 small cubes. Each small cube has atoms at alternate corners as shown in the figure. In all, each small cube has 4 atoms. When these are joined to one another, they make a regular tetrahedron. Thus, there is one tetrahedral void in each small cube and 8 tetrahedral voids in total. Each of the eight small cubes has one void in one unit cell of ccp structure. We know that ccp structure has four atoms per unit cell. Thus, the number of tetrahedral voids is twice the number of atoms.
New answer posted
10 months agoContributor-Level 10
1. When any atom is surrounded by six atoms it creates an octahedral void. In fcc, body centre is surrounded by six atoms present at the face centre. And hence one octahedral void is present at the body centre of each unit cell.
No. of octahedral void at the centre of 12 edge = 12 * = 3
No. of octahedral void at body centre = 1
Total no. of octahedral void in ccp = 3+1+4
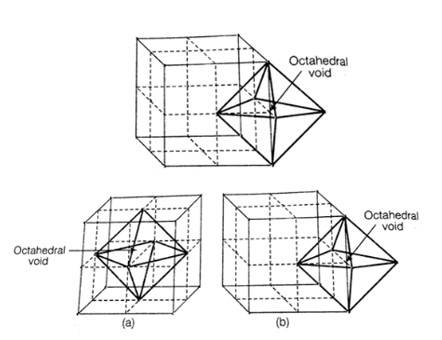
Location of octahedral voids per unit cell of ccp or fcc lattice (a) at body centre of the cube and (b) at the centre of each edge (only one such void is shown)
New answer posted
10 months agoContributor-Level 10
(i) Due to inert pair effect Pb2+ is more stable than Pb4+. Whereas Sn4+ is more stable than Sn2+.
(ii) 3Ga+ —–> 2Ga + Ga3+
This is because Ga3+ is more stable than Ga+
New answer posted
10 months agoContributor-Level 10
| Fullerenes are the allotropes of carbon. Its structure is like a soccer ball. They are prepared by heating graphite in electric arc in presence of inert gases such as helium or argon. |
New answer posted
10 months agoContributor-Level 10
The molecules of CO2 are held together by weak van der Waals forces of attraction which can be easily overcome by collisions of the molecules at room temperature. Consequently, CO2 is a gas.
On the other hand, silicon atom forms four single covalent bonds with O-atom which are tetrahedrally arranged and form a three-dimensional structure. Thus, SiO2 is a high melting solid.
New answer posted
10 months agoContributor-Level 10
Carbon dioxide can be obtained as a solid in the form of dry ice by allowing the liquified CO2 to expand rapidly.
Unlike ordinary ice it does not melt and hence does not wet the surface on which it is kept. Thus, it is called dry ice.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers
