Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
11.4 In the Grignard reagent reaction, the first step of the reaction is the nucleophilic addition of Grignard reagent to the carbonyl group to form an adduct, Hydrolysis of adduct results in the formation of alcohol.
Here, is the general reaction with Grignard reagent below:-
From here, it is clear that HCHO gives CH2OH groups, so R of Grignard reagent is the remaining part of given alcohols. Thus, select the suitable Grignard reagent by substituting the value of R. Now we can see the reaction given below:-
Methanal reacts with iso-propyl magnesium bromide, in presence of dry ether gives an additional compound. And this additional compou
New answer posted
11 months agoContributor-Level 10
11.3 (i) 3-chloroethyl-2-isopropylpentan-1-ol
(ii) 2,5-Dimethylhexane-1,3-diol
(iv) 3-Bromocyclohexanol
(v) Hex-1-en-3-ol
(vi) 2-Bromo-3-methylbut-2-en-1-ol
New answer posted
11 months agoContributor-Level 10
3.60. In a multi-electron atom, the electrons present in the inner shells shield the electrons in the valence shell from the attraction of the nucleus. They act as a screen between the nucleus and these electrons. This is known as the shedding effect or the screening effect.
As the screening effect increases, the effective nuclear charge decreases. Consequently, the force of attraction by the nucleus for the valence shell electrons decreases, and hence the ionization enthalpy decreases.
New answer posted
11 months agoContributor-Level 10
11.2 Allylic alcohol is an organic compound which has the structural formula CH2 = CHCH2OH. In other words, in these alcohols, the-OH group is attached to sp2 hybridized carbon next to the carbon-carbon double bond, that is to an allylic carbon. Therefore, in the above examples, the following are the allylic alcohols.
(ii) H2C = CH – CH2OH and
New answer posted
11 months agoContributor-Level 10
3.59. The d-block elements are known as transition elements.
They have electronic configuration= (n – 1) d1-10 ns1-2
Characteristics of d-block elements are:
They show variable oxidation states.
Their compounds are generally paramagnetic.
Most of the transition elements form coloured compounds.
They are all metals with high melting and boiling points.
These elements are used in the formation of alloys.
New answer posted
11 months agoContributor-Level 10
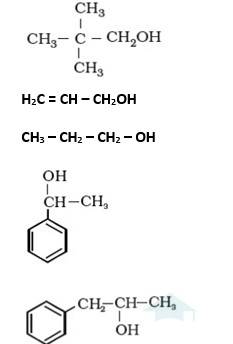
It is primary alcohol because carbon which carries the –OH group is only attached to one alkene group.
It is primary alcohol because the carbon which carries the –OH group is only attached to one propyl group.
It is secondary alcohol because the carbon which carries the –OH group is joined directly to methyl and benzene.
It is secondary alcohol because the carbon which carries the –OH group is joined directly to two different alkyl groups.
New answer posted
11 months agoContributor-Level 10
3.58. Elements gallium and germanium were unknown at the time Mendeleev published his Periodic Table. He left the gap under aluminium and a gap under silicon, and called these elements Eka-Aluminium and Eka-Silicon. Mendeleev predicted not only the existence of gallium and germanium but also described some of their general physical properties.
New answer posted
11 months agoContributor-Level 10
3.57. Ionisation enthalpy depends on the following factors:
1. Atomic size: The ionization enthalpy decreases with an increase in atomic size because with the increase in the atomic size, the number of electron shells increases. Therefore, the force that binds the electrons to the nucleus decreases.
2. Nuclear charge: The ionization enthalpy increases with the increase in the magnitude of the nuclear charge. This is because as the magnitude of the positive charge on the nucleus of an atom increases, the attraction with the electrons also increases.
3. Screening or shielding effect: Greater the magnitude of the screening effect less w
New answer posted
11 months agoContributor-Level 10
3.56. The elements of the s-block and p-block are collectively called as representative or main group elements. These include elements of group I (alkali metals), group 2 (alkaline earth metals).
New answer posted
11 months agoContributor-Level 10
3.55. The energy which is released by an atom in gaining an electron from outside the atom or ion to form a negative ion (or an anion) is called electron gain enthalpy.
The unit of electron gain enthalpy is kJ/ mol. In some cases, like in noble gases, atoms do not have any attraction to gain an electron. In that case, energy must be supplied.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers
