Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoContributor-Level 10
Sign conventions are important because all kinematic variables can be positive or negative. You must first choose your coordinate system and positive direction, then consistently apply signs. For example, if you are choosing upward as positive in free-fall problems, gravity becomes negative (a = -g), upward initial velocity is positive, and downward displacement is negative. The equations of motion work for any situation, as long as you substitute values with proper signs. Incorrect conventions lead to wrong answers.
New question posted
7 months agoNew answer posted
7 months agoContributor-Level 10
It's not always true. Direction of velocity is the most important consideration here that will tell us whether acceleration increases or decreases speed. You can consider two scenarios. If you're falling, that's negative velocity. That implies negative gravitational acceleration. Here, your speed increases. If you're moving upward, that's positive velocity. Now, with that same negative acceleration, your speed decreases. For choosing the equations of motion, you need to know both the signs of acceleration and velocity to determine if you're speeding up or slowing down.
New answer posted
7 months agoContributor-Level 10
Zero velocity doesn't mean zero acceleration. When you throw a ball-like object upward, at its peak the velocity is zero. But acceleration remains constant due to gravity. The velocity is still changing from positive to negative at that instant. That means acceleration continues.
New answer posted
7 months agoContributor-Level 10
Reduced pressure distillation technique is used for purification of high boiling organic liquid which decompose near its boiling point. By reducing pressure they boil below their normal boiling point.
New answer posted
7 months agoContributor-Level 10
Solubility of gas in liquid increases on decreasing the temperature. So water at 4°C will have more dissolved O2.
New answer posted
7 months agoContributor-Level 10
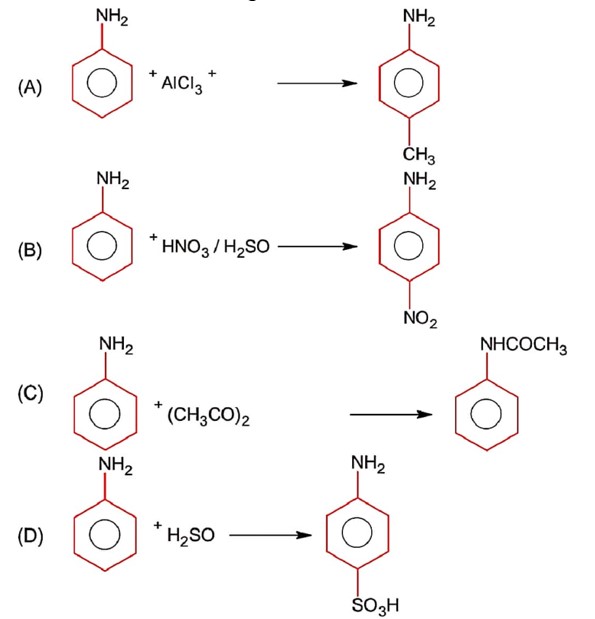
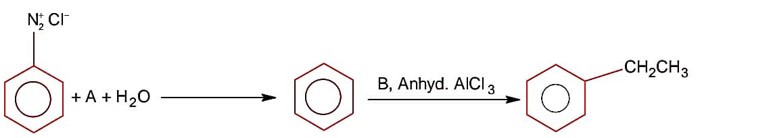
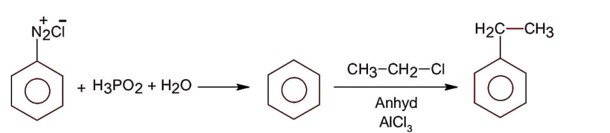
Aniline being a base does not give Friedel Craft reaction because it react with AlCl3 used in Friedel Craft reaction.
New answer posted
7 months agoContributor-Level 10
Ba has outer electronic configuration 6s2.
CaC2O4 is insoluble in water.
Compound of Li are covalent so soluble in organic solvent.
Na forms strong monoacidic base.
New answer posted
7 months agoContributor-Level 10
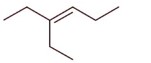
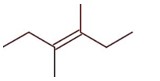
3-Ethylhex-3-ene will not show stereo isomerism
Methylhex-1-ene will show steroisomerism (optical isomerism)
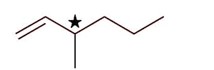
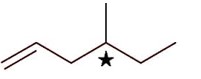
4-Methylhex-1-ene will show stereoisomerism (optical) since it has one chiral carbon
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers