Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
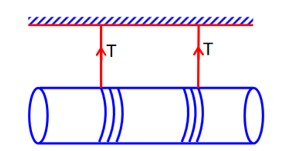
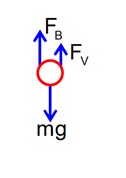
m = 0.3g density,
Fv + FB = mg.
=Fv = mg – FB = .3 * 10-3 * 10 – 1.3 *
= 2.5 * 10-3N
= 25 * 10-4N
x = 25
New answer posted
8 months agoContributor-Level 10
Higher melting point means more energy is required to break the ionic bond. Since the ionic bonds in MgO are stronger than in NaCl due to various reasons, It have higher melting and even boiling point. Reasons are listed below:
- In MgO, Magnesium and oxygen ions carry a +2 and -2 charge while sodium and in NaCl, chlorine ions carry a +1 and -1 charge. Due to higher charges more electrostatic attraction is involved.
- The smaller ionic radius of Mg2+ are smaller compared to Na+, makes the bond stronger in MgO.
- Due to larger elctrostatic force and smaller ionic radius, ionic bond in MgO are stronger, resulting in higher lattice energy.
- The stro
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers