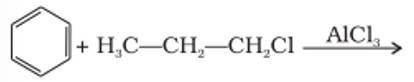Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
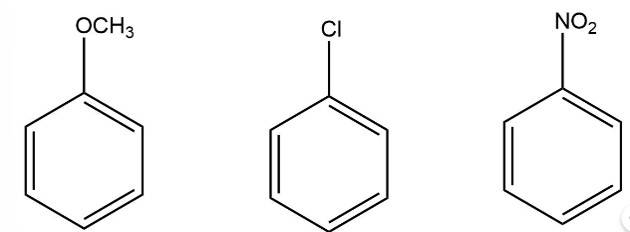
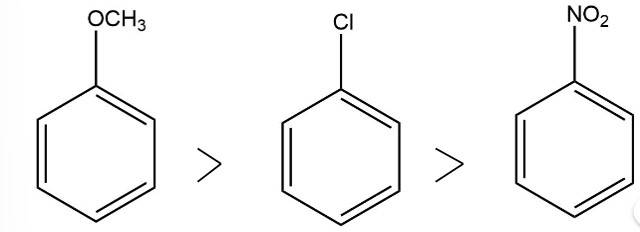
The electron donating group increases the reactivity in electrophilic substitution reaction whereas the electron withdrawing group decreases the reactivity in electrophilic substitution.
Thus, the order of reactivity
New answer posted
9 months agoContributor-Level 10
ν = 4.37 x 105 m s-1, m = 0.1 kg
As per de Brogile's equation,
λ= m/v = (6.626 x 10-34 kg m2 s-1) / (0.1 kg) x (4.37 x 105 m s-1)
=6.626/0.437 x 10-34-5 m
= 1.516 x 10-38 m
New question posted
9 months agoNew answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
New answer posted
9 months agoContributor-Level 10
ν = 2.19 x 106 m s-1
As per de Brogile's equation,
λ= m/v = (6.626 x 10-34 kg m2 s-1) / (9.1 x 10-31 kg) x (2.19 x 106 m s-1)
6.626/91 * 2.19= x 10-34+25 m = 0.33243 x 10-9 m = 332.43 pm
New answer posted
9 months agoContributor-Level 10
λ = 800 pm = 800 x 10-12 m
m = 1.675 x 10-27 kg
As per de Brogile's equation,
ν =h/mλ = (6.626 x 10-34 kg m2 s-1) / (1.675 x 10-27 kg) x (800 x 10-12 m)
=6.626/1.675 * 8 x 10-34+27+10
= 0.494 x 103 ms-1
= 494 ms-1
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
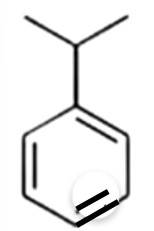
The benzene here would undergo Friedel-Crafts alkylation reaction where carbocation is formed as an intermediate, thus secondary carbocation is formed as an intermediate due to its greater stability than that of the primary carbocation.
The final product obtained is
New answer posted
9 months agoContributor-Level 10
As per de Brogile's equation,
λ = h / mv = (6.626 x 10-34 kg m2 s-1) / (9.1 x 10-31 kg) x (1.6 x 106 ms-1)
= 0.455 x 10-9 m = 0.455 nm = 455 pm.
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The rate-determining step involved in the reaction is
CH3-CH=CH2 + HX → CH3-CH+-CH3 + X-
So the rate-determining step depends on the bond energy of HX i.e. higher the bond energy lesser would be reactivity.
Thus the order of reactivity of these halogen acids is
HI>HBr>HCl
New answer posted
9 months agoContributor-Level 10
Radius of orbit of H like species = (0.529 / Z) n2Å = (52.9 / Z) n2 pm
r1 = 1.3225 nm = 1322.5 pm = (52.9 / Z) n12
r2 = 211.6 pm = 211.6 pm = (52.9 / Z) n22
∴ r1 / r2 = 1322.5 / 211.6
=>n12 /n22 = 6.25
=> n1/n2= (6.25)1/2 = 2.5
=> n1 = 2.5 n2
=> 10 n1= 25 n2
=> 2 n1= 5 n2
If n1 = 2, then n2 = 5. That means transition occurs from 5th orbit to 2nd orbit. This means that the transition belongs to Balmer series.
Now, wave number? = (1.097 x 107 m-1) x (1/22 – 1/52) = 1.097 x 107 x 21/100 m-1 = 23.037 x 105 m-1
λ = 1/? = 1/ 23.037 x 105 m-1
= 434 x 10-9 m = 434 nm
This transition belongs to visible region of the spectrum of light.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers