Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
6 months agoContributor-Level 10
The slag formed is CaSIO3 which is lighter and has less melting point.
New answer posted
6 months agoNew answer posted
6 months agoContributor-Level 10
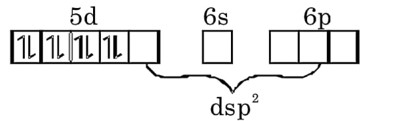
(a) [1rCl (CO) (PPh3)2] carbonylchloridobis (triphenylphosphine) iridium (I).
(b) Coordination number of Ir is four. Ir is in (+1) oxidation state with 4d8 configuration. It is trans isomer, so its geometry should be square planar.
(c) All electrons are paired, hence diamagnetic
(d) Can exhibit GI only.
New answer posted
6 months agoContributor-Level 10
Fe acts a catalyst and Mo acts as promoter in Haber's process.
New answer posted
6 months agoContributor-Level 10
BOD value less than 5ppm is considered as clean water and BOD value more than 10ppm is considered as polluted water.
New answer posted
6 months agoContributor-Level 9
Reaction rate is used to measure how fast or slow reactions occur per unit time. The rate constant is a proportionality factor that remains constant for every reaction.
New answer posted
6 months agoContributor-Level 9
Yes, in elementary reactions, order and molecularity can be the same, but this is not always the case because order is an experimental quantity, and molecularity is a theoretical concept.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers