Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
6 months agoContributor-Level 10
The reaction is X²? → Y¹²? + Z¹²?
Binding energies per nucleon are: X=7.6 MeV, Y=8.5 MeV, Z=8.5 MeV.
Gain in binding energy (Q) = (Binding energy of products) - (Binding energy of reactants)
Q = (120 * 8.5 + 120 * 8.5) - (240 * 7.6) MeV
Q = (2 * 120 * 8.5) - (240 * 7.6) MeV = 2040 - 1824 = 216 MeV.
New answer posted
6 months agoContributor-Level 10
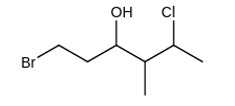
The IUPAC name giving the lowest number to the functional group obeying the lowest set of locants is 1-bromo-5-chloro-4-methylhexan-3-ol.
New answer posted
6 months agoContributor-Level 10
More the number of strong field ligands in a complex greater the energy absorbed by a complex.
New question posted
6 months agoNew answer posted
6 months agoContributor-Level 10
Oxidation state in neutral medium changes from +7 to +4 for MnO? to MnO?
New answer posted
6 months agoContributor-Level 10
In n-type semiconductor majority charge carriers are e- and P type semiconductor majority charge carriers are holes.
neA
New answer posted
6 months agoContributor-Level 10
Ecell = 1.05 - (0.059 / 2) log ( [Ni²? ] / [Ag? ]²)
= 1.05 - (0.059 / 2) log ( [10? ³] / [10? ³]²)
= 1.05 - (0.059 * 3) / 2 = 1.05 - 0.0885 = 0.9615 volt
There is a misprint in the question. The E? cell is incorrectly given as 10.5 V. This should have been 1.05 volt.
New answer posted
6 months agoContributor-Level 10
Statement I is true but statement II is incorrect as 3°-alcohols are the most reactive and give immediate turbidity with Luca's reagent.
New answer posted
6 months agoContributor-Level 10
Hematite: Fe? O?
Magnetite: Fe? O?
Calamine: ZnCO?
Kaolinite: [Al? (OH)? Si? O? ]
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers