Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoContributor-Level 10
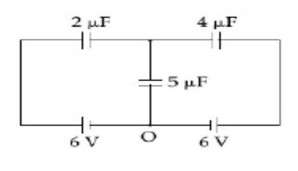
Now, using junction analysis
We can say, q? + q? + q? = 0
2 (x - 6) + 4 (x - 6) + 5 (x) = 0
x = 36/11, q? = 36 (5)/11 = 180/11
q? = 16.36µC
New answer posted
7 months agoContributor-Level 10
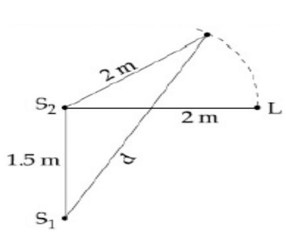
Initially S? L = 2m
S? L = √2² + (3/2)²
S? L = 5/2 = 2.5 m
? x = S? L - S? L = 0.5 m
So since λ = 1 m. ∴? x = λ/2
So white listener moves away from S? Then? x (= S? L − S? L) increases and hence, at? x = λ first maxima will appear.? x = λ = S? L − S? L.
1 = d - 2 ⇒ d = 3 m.
New answer posted
7 months agoContributor-Level 9
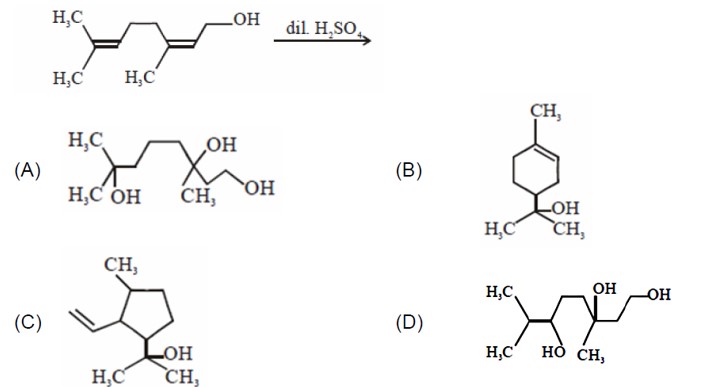
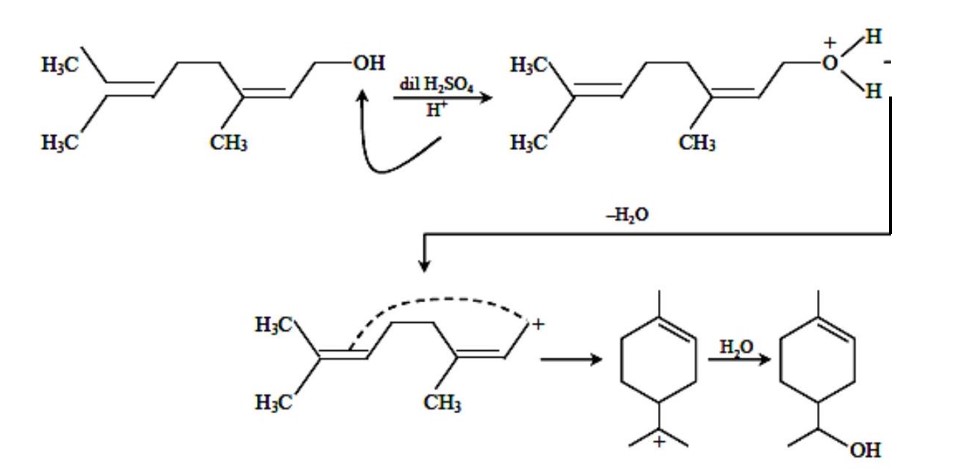
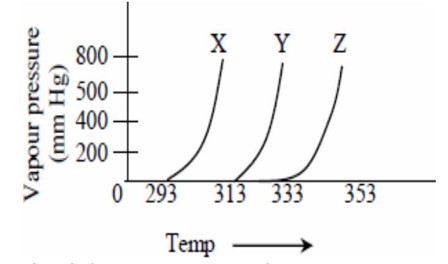
At a fixed temperature, having more vapour pressure as compared to . So, intermolecular interaction is lower as compared to .
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers