Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: B
Hydrate isomerism is isomerism in which water is used as a solvent.
Coordination compounds with the same composition but varying ligand connectivity are known as linkage isomers.
Solvate isomerism has the same composition as free solvent but distinct solvent ligand molecules.
Except for the ligand that swaps places with the anion, ionisation isomers are identical isomers.
Coordination isomers are coordination compounds with distinct metal and ligand compositions.
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: B
Ligands are neutral ions that form a coordination complex with the central metal atom.
NH4+ does not have a lone pair of electrons to give.
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: A
A chelating agent binds to a single metal ion with two or more donor atoms. Oxaloto, Glycinato, and ethane-1,2-diamine all have two donor oxygen atoms. The gland Thiosulpato is ambidentate.
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: D
Two or more compounds with the same formula but distinct structures and characteristics are known as isomers.
So, in this case compounds
[Co (SO4) (NH3)5]Br and [Co (SO4) (NH3)5]Cl show no isomerism
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: (A)
Linkage isomerism arises in a coordination compound containing ambidentate ligand.
A simple example is by complexes containing the thiocyanate ligand, NCS-which may bind through the nitrogen to give M-NCS or through sulphur to give M-SCN.
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: A
CFSE for octahedral and tetrahedral complex is related as
Δt = Δ0
Where Δ0= CFSE for octahedral complex
Δ0= CFSE for tetrahedral complex
Δ0=1800 cm−1
Δt =49*18000=8000 cm−1
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: A
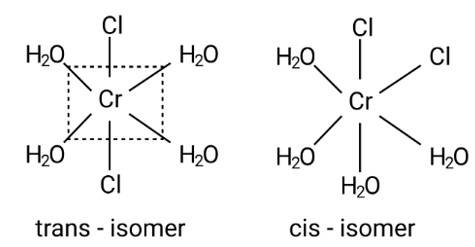
This is due to the complex's octahedral structure and (cis-trans) isomerism. Two Cl ligands are next to each other in trans isomerism. Although these isomers have the same structure, they are not identical.
Possible isomers are:
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: C
The chelate effect is when a chelating ligand coordinates the stabilisation of a molecule. Any ligand that binds to metal forms a ring with 5 or 6 members, which is the most stable.
Here, CO, CN, H2O is monodentate ligand [Fe (C2O4)3]3- is an oxalate ligand.
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: A
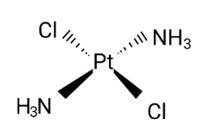
Diamminedichloridoplatinum (II) is also known as azane
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: D
When 1 mol [CrCl3 (H2O)3]6H2O is treated with excess of AgNO3, 3 mol of AgCl is produced, i.e., [CrCl3 (H2O)3]6H2Ois dissociated in aqueous solution and all three chloride comes in solution.
[Cr (H2O)6]Cl3→ [Cr (H2O)6]3+ + 3Cl-
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers
