Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
39. (A) 25
2MnO4- + 5S2- + 16H+ → 2Mn2+ + 5S + H2O
From the reaction,
For 5 moles of S, two moles of KMnO4 were required.
Therefore, for one mole of S, the number of moles of KMnO4 required will be = 25.
New answer posted
10 months agoContributor-Level 10
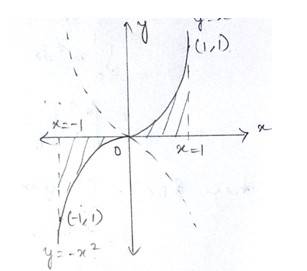
Given curve is
for
And
We know that at i.e,
So the point of intersection is at

New answer posted
10 months agoContributor-Level 10
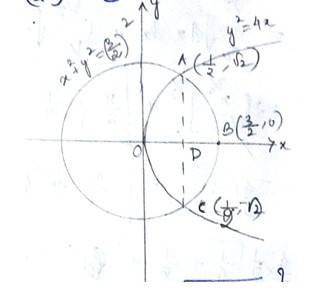
The given area of the circle is is a circle with centre (0,0) and radius, and the parabola is -------------(2)
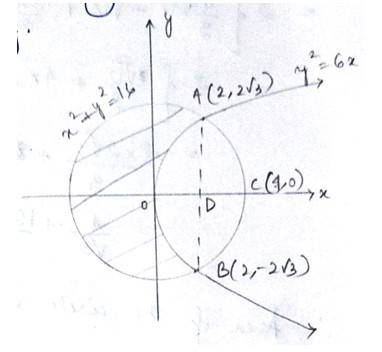
Solving (1) and (2) for x and y.
For,
Which is not possible.
For,
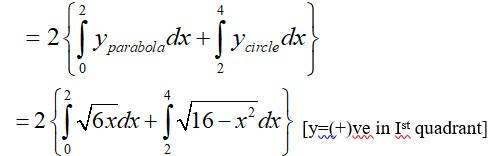

New answer posted
10 months agoContributor-Level 10
38. (C) Tm
In the periodic table, actinides lie from atomic number 90 to 103.
Thulium is not an actinide; it is a lanthanide.
New answer posted
10 months agoContributor-Level 10
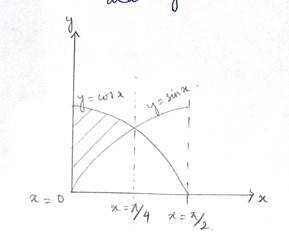
The given curve is
Which is in the form of a parabola nad the lines are
At
At
Shaded area of the Ist quadrant
Shaded area of the IInd quadrant
Total area of the enclosed region
Option (c) is correct.
New answer posted
10 months agoContributor-Level 10
37. (D) Mn2+ acts as auto catalyst
Mn2+ formed in the reaction acts as an autocatalyst. Auto catalyst is a compound that is one of the products that are formed in the reaction itself catalyses the reaction.
Reaction: 2MnO4- + 16H+ + 5C2O42- → 2Mn2 + + 10CO2 + 8H2O
New answer posted
10 months agoContributor-Level 10
55. (ii) Both assertion and reason are true but reason is not the correct explanation of assertion.
Explanation: Hydrometallurgy is used to extract copper from low-grade ore. Hydrometallurgy entails dissolving the ore in a suitable reagent and then precipitating it. In this method, more electropositive metal is used, allowing pure metal to be displaced.
New answer posted
10 months agoContributor-Level 10
36. (A)i, ii
Disproportionation/redox reaction is a reaction in which one compound is oxidised as well as reduced at the same time.
(A) Cu+ ? Cu2+ + Cu
( + 1) ( + 2) (0)
(B) 3MnO4- + 4H+ ? 2MnO4- + MnO2 + 2H2O
( + 6) ( + 7) ( + 4)
New answer posted
10 months agoContributor-Level 10
54. (ii) Both assertion and reason are true but reason is not the correct explanation of assertion.
Explanation: The zone refining method is very useful for producing high-purity semiconductors and other metals, such as germanium.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers