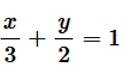Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
Given equation of the curve is , which can be break down into each quadrant .
For Ist quadrant,
i.e., - (1)
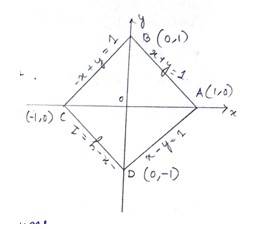
Similarly for IInd, IIIRd nad IVth quadrant
- (2)
- (3)
- (4)
We draw the above focus lines on a graph and find the area enclosed which is a square.
Required area .
New answer posted
10 months agoContributor-Level 10
The given equation of the parabola is ---------(1)
and that the line is --------------(2)
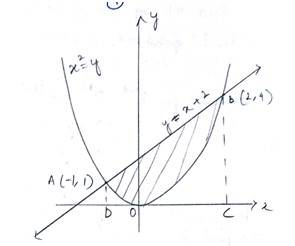
Solving (1) and (2) for x and y
When
And
The point of intersection of the parabola and the lines
Hence the required area enclosed region is
New answer posted
10 months agoContributor-Level 10
50. Option (i) A (2) B (3) C (4) D (1)
Explanation: A solidified copper has a blistered appearance due to the evolution of SO2 Hence it is called blistered copper.
Hence, option (A) from column I is matched with option (2) from column II. Iron is extracted from a blast furnace.
Hence, option (B) from column I is matched with option (3) from column II. The iron ore is heated in the reverberatory furnace after mixing with silica. In the furnace, iron oxide slags of iron and copper are produced in the form of copper matte.
Hence, option (C) from column I is matched with option (4) from column II. The hall-Heroult process is used for the extra
New answer posted
10 months agoContributor-Level 10
32. (B) CuF2
(A) Ag2SO4 (Ag+ )→ 5d106s0
(B) CuF2 (Cu2+ )→ 3d94s0
(C) ZnF2 (Zn2+)→ 3d104s0
(D) Cu2Cl2 (Cu+)→ 3d106s0
Unpaired electrons present in any compound impart colour to the salt of transition metal. Only CuF2 has unpaired electrons in its 3d orbital that's why it is white coloured in its solid-state while rest of the salts are colourless.
New answer posted
10 months agoContributor-Level 10
The Given equation of the ellipse is
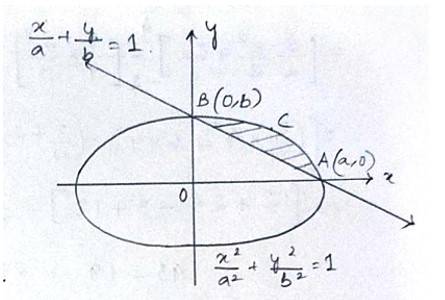
And the equation of the line in
With x and y intersept a and b
So, required area of the enclosed region is
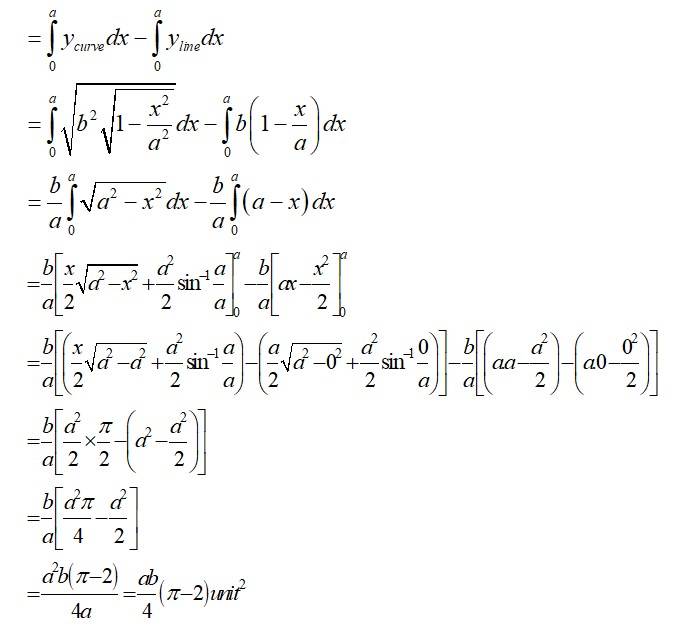
New answer posted
10 months agoContributor-Level 10
Given equation of the ellipse is Which as major axis aling x- axis and that of the line is which has x and y intercepts at 3 and 2respectively.
Required area of enclosed region is area
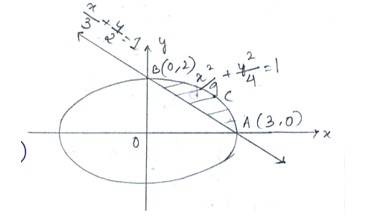
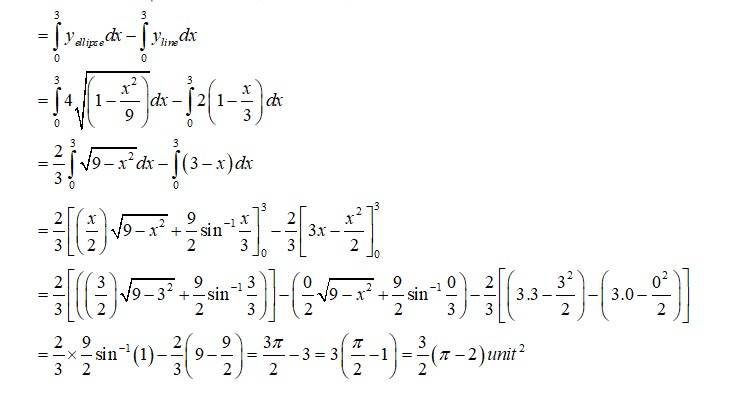
New answer posted
10 months agoContributor-Level 10
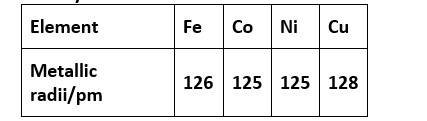
31. (D) Cu
Density is mass by volume. As we move from left to right for a long period, the atomic radii decrease. Hence, volume decreases. Also, an increase in atomic masses is observed.
So overall the density increases out of the above option from iron to copper, copper will have the highest density.
New answer posted
10 months agoContributor-Level 10
The given equation of parabola is ------------(1)
And the line is ----------------------(2)
Solving (1) and (2) for x and y,
At,
And
Thus, the point of intersection of (1)&(2)are
Area of the enclosed region (BOAB)
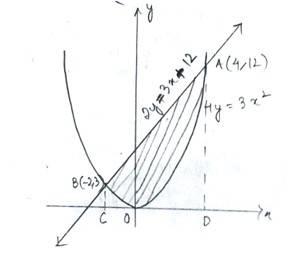
=area (CBAD) – area (OADC)
New answer posted
10 months agoContributor-Level 10
49. Option (i) A (3) B (4) C (2) D (1)
Sapphire is a gemstone containing Co.
Hence, option (A) from column I is matched with option (3) from column II. The Sphalerite single is ZnS.
Hence, option (B) from column I is matched with option (4) from column II. NaCN is also used as a depressant.
Hence, option (C) from column I is matched with option (2) from column II. Al2O3 is also called corundum.
Hence, option (D) from column I is matched with option (1) from column II.
New answer posted
10 months agoContributor-Level 10
30. (A) Cu (II) is more stable .
Electronic configuration of Cuis [Ar] 3d10 4s1
Cu (I) - [Ar] 3d10 4s0
Cu (II)- [Ar] 3d9 4s0
Despite the fact that Cu (I) has fully filled 3d-orbital but Cu (II) is more stable than Cu (I) due to the greater effective nuclear charge of Cu (II) as nucleus has to hold 17 electrons rather than 18 electrons like in Cu (I).
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers

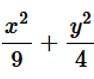 and the line
and the line