Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
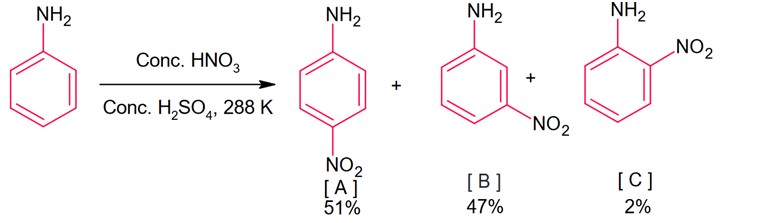
During nitration of aniline, meta product is also formed, it is because of due to presence of anilinium ion. In anilium group is meta directing group.
New answer posted
9 months agoContributor-Level 10
(a) Na2CO3 -> Solvay
(b) Ti -> Van-Arkel
(c) Cl2 -> Deacon
(d) NaOH -> Castner – kellner
New answer posted
9 months agoContributor-Level 10
A > B > C > D
Lone pair is localized in (A) while all 3 have delocalized lone pair but they can be compared by 3° > 2° > 1° because methyl group increases the basicity.
New answer posted
9 months agoContributor-Level 10
Both (A) and (B) are correct but R is not correct explanation of A. In both
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers