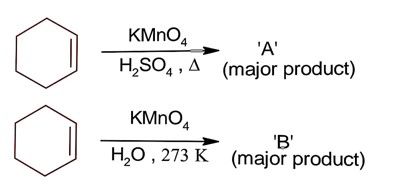
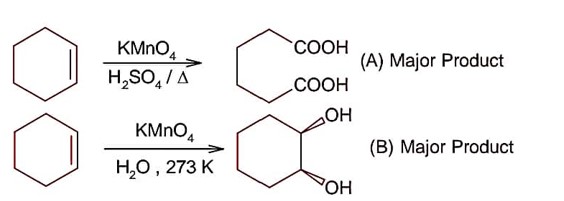
Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
Depression in freezing point will be maximum for Al2 (SO4)3 since its Van't Hoff factor is the highest. So its solution will have the lowest freezing point.
New answer posted
9 months agoContributor-Level 10
When AgNO3 is added to Kl solution, precipitate of Agl is formed which adsorb I- ion from
Dispersion medium to give negatively charged sol
Agl/l-- negatively charged sol.
New answer posted
9 months agoContributor-Level 10
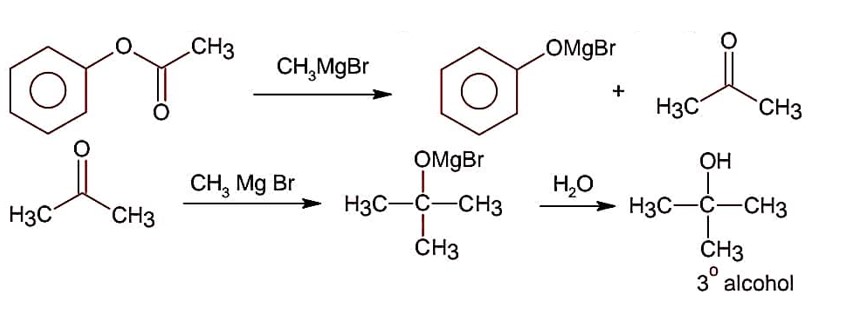
Esters on treating with excess CH3MgBr followed by hydrolysis gives 3° alcohol.
New answer posted
9 months agoContributor-Level 10
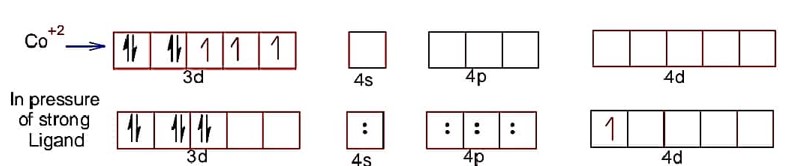
Hence, electronic configuration of CO2+ is
spin magnetic moment =
New answer posted
9 months agoContributor-Level 10
Both statement are correct for the glass body heating, but reason is not correct explanation during heating process of glass, constituents unit rupture of glass body and gives the edge smoothness.
New answer posted
9 months agoContributor-Level 10
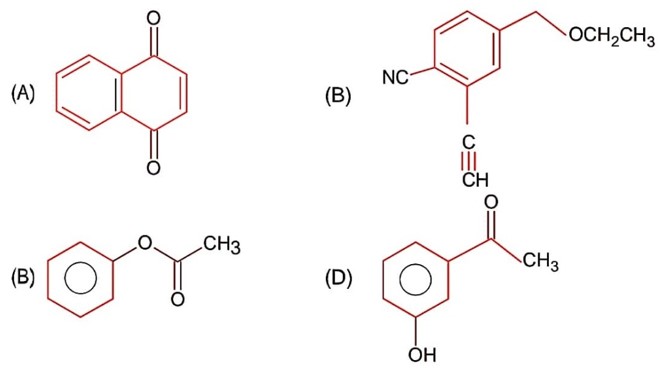
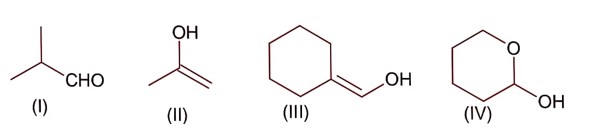
All structure can produce -CHO functional group which gives +ve test of Tollen's reagent except structure (II), because if forms ketonic group after tautomerisation
New answer posted
9 months agoContributor-Level 10
Deep blue colour solution
In cation analysis of Cu+ ions, precipitate formed is CuS on treating with H2S and HCl which dissolved in HNO3 and produced blue colour complex solution
New answer posted
9 months agoContributor-Level 10
In
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers