Biomolecules
Get insights from 85 questions on Biomolecules, answered by students, alumni, and experts. You may also ask and answer any question you like about Biomolecules
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
Correct option is C.
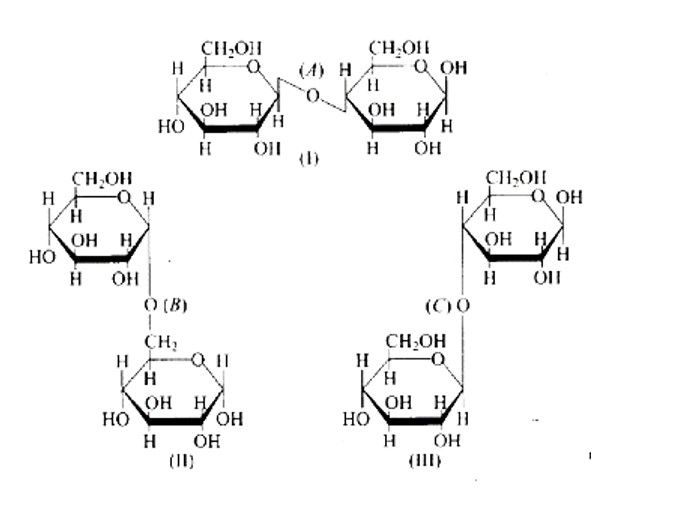
The links between C1 and C4 of glucose are shown as 'A' and ' C, ' respectively, whilst the linkage between C1 and C6 of the glucose units is shown as ' B. ' Furthermore, option (c) is clearly correct based on the structures.
Because ' B ' is the bond or link between the glucose units of C1 and C6 and, option (b) is wrong.
Option (a) is wrong because the relationship between C1 and C4 and is represented by 'C.'
Because 'A' is the hyperlink between C1 and C4 the glucose units of and, option (d) is wrong.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
The Correct option is c.
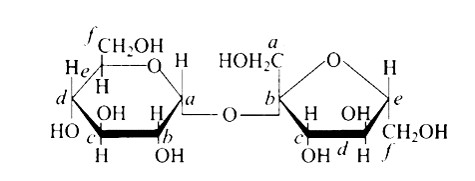
In the cyclic structure of glucose or fructose, anomeric carbon is carbon that is close to an oxygen atom. As shown in the structure above, atoms 'a' and 'b' are close to the oxygen atom, and the hydroxyl groups of both carbon atoms have different orientations.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
The Correct option is a.
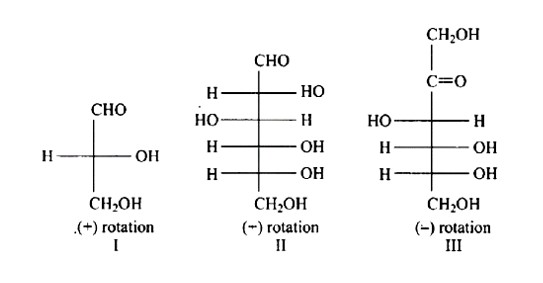
In the same way as (+) glyceraldehyde has a group on the lowest asymmetric carbon on the right side, the I, II, and III structures have a (-OH) group on the lowest asymmetric carbon on the right side giving them a D-configuration.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
The Correct option is c.
The absence of a free (-CHO) aldehyde group is indicated by the fact that glucose pentaacetate does not react with hydroxylamine. The open structure of glucose cannot account for this, although the open chain structure of glucose can account for all other features.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
Ans: The Correct option is a.
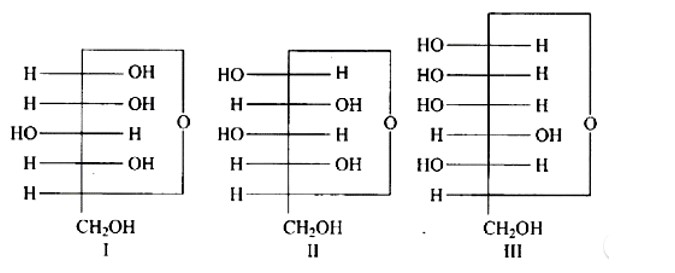
Anomers are cyclic monosaccharide structures that differ structurally at carbon-1. In this scenario, I and II are anomers since they differ only at carbon -1.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
The Correct option is d.
Adenine, guanine, thymine, and cytosine are the four bases found in DNA. As a result, uracil is not found in DNA as we cannot identify it among the other nucleotides that are present in the DNA. Uracil is, however, seen in RNA, another nucleic acid found in most of the organisms.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
The Correct option is d.
Water-soluble vitamins are those in the B group. B group vitamins cannot be stored in our systems since they are quickly excreted; but, because they are B12 insoluble in water, our bodies may store the vitamin. Because our systems are unable to store these vitamins, options (a), (b), and (c) are excluded.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
The Correct option is c.
The four bases contained in DNA are adenine, guanine, thymine, and cytosine. Adenine, uracil, guanine, and cytosine are the four bases of RNA. As a result, RNA does not include thymine as in place of thymine, it has uracil which is a lesser stable nucleotide base and is somewhat responsible for RNA's instability but also lends it some flexibility to change.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
The Correct option is a.
Proteins contain one or more polypeptide chains. When each polypeptide of a protein's amino acids is joined in a precise order, a proteins' main structure is referred to as its primary structure. This one is the linear structure with no bonds other than the ones between adjacent amino acids. The sequence of amino acids in the primary structure is pre-determined by the genetic code of an individual and thus is a specific sequence.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
The Correct option is c.
The pyranose form of glucose, which has a five-membered ring structure, is found. As a result, option (c) is wrong.
Glucose is a six-carbon monosaccharide with an aldehyde group. As a result, it's referred to as an aldohexose. As a result, the statement in option (a) is correct.
When glucose is cooked with HI, n-hexane is produced. All six carbon atoms in glucose are linked together in a straight chain during this procedure. As a result, the statement in option (b) is correct.
The aldehyde group is not free in the cyclic structure. As a result,
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 690k Reviews
- 1850k Answers