Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(IV) o-methoxy phenol
The stability of the conjugate base determines the acidic character of the compound. Higher the stability of the conjugate base, the higher is the acidic character.
Phenol, O-nitrophenol, O-methyl phenol, and O-methoxy phenol all are aromatic compounds. In aromatic compounds, the negative charge of the conjugate base charge is delocalised. The delocalisation of negative charge gives additional stability to the compound.
Ethanol is not aromatic and it cannot delocalise the negative charge formed after the H+ ion leaves the c. So,
New question posted
9 months agoNew answer posted
9 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(I) C6H5OH
C6H5OH, phenol is a strong acid (due to resonance) which will react with sodium hydroxide solution in water to form sodium phenoxide and water. Phenol is more acidic than sp3 hybridized attached alcohols.
New answer posted
9 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
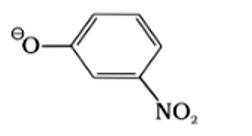
(II) ΘOR
The species which is the weakest acid has the strongest conjugate base. The ROH is the weakest acid among the given compounds due to the inductive effect of the alkyl group. Therefore, the strongest conjugate base is ΘOR.
New answer posted
9 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
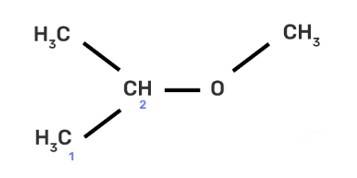
(II) 2-methoxy-2-methylethane
IUPAC provides consistency to the names of organic compounds. It enables every compound to possess a unique name, which otherwise is not plausible with the common names. The chemical structure of the given compound is shown below:
The given compound is an alkane so suffix in our case will be –ane. An ether functional group is also present so prefix will be Alkoxy-.
To determine the name of a compound, identify the longest and continuous chain of carbon containing the functional group and count the number of carbon atoms in the chain as shown i
New answer posted
9 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
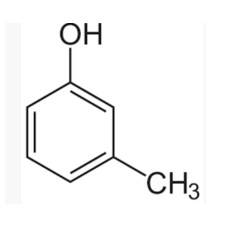
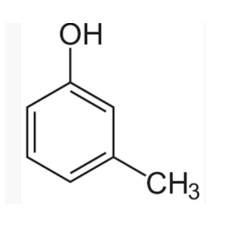
(I) 3-methylphenol
The m-cresol is an organic compound which contains a benzene ring, a -OH group and a methyl group. It has a methyl group substituted to meta position in the phenol ring. According to the IUPAC system, the -OH group is given more priority than the methyl group.
The cresols are also called hydroxytoluene or the methylphenols. These contain a benzene ring with one methyl and one phenol group substituted. There are three forms of cresols which are o-cresol, m-cresol and p-cresol. m- cresol is a colourless liquid and is viscous in nature.
In case of m-cre
New answer posted
9 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(III) 5-Chlorohexan-2-ol
The IUPAC naming of the given compound are as follows:
Word root: It depends upon the number of carbon atoms in the longest continuous carbon chain selected, called the parent chain. Depending upon the number of carbons in the chain the compound is assigned a word. The given compound contain 6 carbon attached in the continuous the longest chain and word root for 6 carbon atoms is 'hex'.
The suffix- A suffix is added after the word root to indicate the nature of the carbon-carbon bond. The given carbon chain contains a single bond then a s
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers