Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
4NH3 + 5O2 ![]() 4NO + 6H2O
4NO + 6H2O
New answer posted
9 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
SO3 generates a dense fog of sulphuric acid that does not condense quickly, it is not absorbed directly in water to form H2SO4.
New answer posted
9 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
A = NH4NO2
B = N2
C = NH3
D = HNO3
(i) NH4NO2→N2 + 2H2O
(ii) N2 + 3H2→2NH3
(iii) 4NH3 + 5O2→4NO + 6H2O
4NO + O2→4NO2
3NO2 + H2O→2HNO3 + NO
New answer posted
9 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
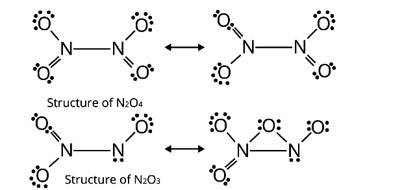
Here, 'A' is NO2 (Nitrogen dioxide)
'B' is N2O4 (dinitrogen tetraoxide)
'C' is N2O3 (dinitrogen trioxide)
A brown gas is produced when lead nitrate (II) is heated
2Pb (NO3)2 ![]() 2PbO + 4NO2 + O2
2PbO + 4NO2 + O2
2NO2 ![]() N2O4
N2O4
2NO + N2O4  2N2O3
2N2O3
New answer posted
9 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
'A' is S8 'B' is SO2 gas
S8 + 8O2 ![]() 8SO2
8SO2
2MnO4 – + 5SO2 + 2H2O → 5 SO42– + 4H+ + 2Mn2+
(violet) (colourless)
2Fe3+ + SO2 + 2H2O → 2Fe2+ + SO42– + 4H+
New answer posted
9 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
option (i)
Explanation: The entropy of a liquid reduces as it crystallises. Because the molecules are more organised in crystalline form.
New answer posted
9 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: (ii)
Explanation: The energy factor for a spontaneous process should be favourable (i.e., -ve) and the randomness should be positive.
New answer posted
9 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
option (ii)
Explanation: The enthalpy of the reactants is always greater than the enthalpy of the product in a combustion reaction.
New answer posted
9 months agoContributor-Level 10
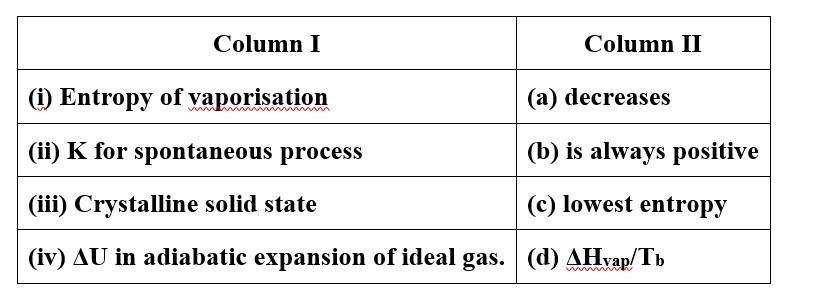
This is a Matching Type Questions as classified in NCERT Exemplar
(i)- (b), (d)
(ii)- (b)
(iii)- (c)
(iv)- (a)
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers