Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
If all gases are at the same temperature and pressure, Gay lussac's law of gaseous volumes states that gases combine or are created in a chemical reaction in a simple volume ratio.
H2 (g) + Cl2 (g) → 2HCL (g)
1 volume 1 volume 2 volume
22.4 litre 22.4 litre 44.8 litre
2N2 (g) + O2
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (iv)
Characteristics of cathode rays depend upon the nature of gas present in the cathode ray tube. As per the result obtained from the cathode ray discharge tube experiment the characteristics of cathode rays (electrons) does not depend upon the material of electrodes and the nature of the gas present in the cathode ray tube. Which concludes that electrons are the basic constituent of all the atoms
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
ANS – Option (iv)
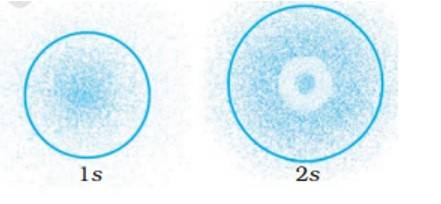
The probability density of electrons for 2s orbital decreases uniformly as distance from the nucleus increases. Electrons at the 1s orbital decreases as we move far from the nucleus, however in case of 2s the probability decreases initially then it increases with the distance and thereafter at a certain point it starts decreasing with the distance.
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Molecular mass of Ca3 (PO4)2 = 310.18 g / mole
Given mass of calcium 4 x 30= 120 g
Given mass of phosphorous = 31 x 2 =62 g
Given mass of oxygen = 16 x 8 = 128 g
Mass percent of Calcium = x 100 = 38.71%
Mass percent of Phosphorous = x 100 = 20%
Mass percent of Oxygen = x 100 = 41.29 %
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (ii) 1s2 2s2 2p6 3s2 3p6 3d9 4s2
As per the Hund's rule the half-filled and fully filled orbital leads to the extra stability due to the symmetry thus fully filled 3d and half-filled 4s is preferred.
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
The number of moles of a substance (known as the solute) dissolved in precisely 1 litce of a solution is known as molarity (solvent and solute combined). As a result, the formula for estimating molarity is as follows:
Molarity =
The term molarity is also used to refer to molar concentration. As a result, molar concentration measurement is based on the volume of liquid in which a substance is dissolved. It's vital to remember that the volume is in litres, so if we have volume in mL we need to convert that in liters.
Molality is the number of moles of substance (also k
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (iii)
Electrons move in a circular path of fixed energy called orbits .
As per Rutherford's α-particle scattering experiment the nucleus is surrounded by electrons that move around the nucleus with a very high-speed in circular paths called orbits. It does not mention the energy or stability of the electrons revolving around the nucleus.
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
SI unit of the mole is mol. The amount of a substance that contains as many particles or entities as there are atoms in exactly
12 g (0.012 kg) of the C-12 isotope is defined as a mole. One mole is defined as follows:
1 mole = 6.023 x 1023
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
On solving the above equation, the result is 5.4. All non-zeroes digits are significant. The significant figure is 2.
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Mass of 1 mole of C-12 = 12g
1 mole contains 6.022*1023 atoms.
Thus, mass of 6.022*1023 atoms=12g
Mass of 1 atom of carbon =126.022*1023 g
=1.99*10−23 g
Thus, mass of one atom of C-12 is 1.99*10−23 g
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers