Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans:
(i) Cr (d) [Ar] 3d54s2
(ii) Fe2+ (c) [Ar]3d64s0
(iii)Ni2+ (a) [Ar]3d84s0
(iv)Cu (b) [Ar]3d104s1
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (C)
The empirical formula mass of CH2O is 30 g
The relation between empirical and molecular formula is given as,
n = …. (1)
On substituting the values in equation (1)
n = = 6
Thus, the molecular formula of the compound will be,
Molecular formula of the compound = (CH2O)6 = C6H12O6
New answer posted
10 months agoContributor-Level 10
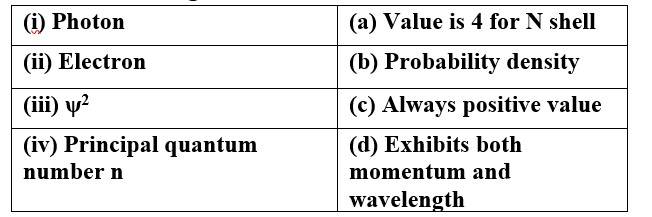
This is a Matching Type Questions as classified in NCERT Exemplar
Ans:
(i) Photon (d) Exhibits both momentum and wavelength
(ii) Electron (d) Exhibits both momentum and wavelength
(iii) ψ 2 (b) Probability density
(c) Always positive value
(iv) Principal quantum (a) Value is 4 for N shell
number n (c) Always positive value
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (B)
The molar mass of carbon is 44 g mol-1
44 g of carbon dioxide contains 12 g of carbon
The percentage composition is given as, % composition
=
On substituting the values in the above equation,
% of carbon =
= 27.27%
New answer posted
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans:
(i) X-rays (d) ν = 1018Hz
(ii) UV (c) ν = 1016Hz
(iii) Long radio waves (a) ν = 10 -104 Hz
(iv)Microwave (b) ν = 1010 Hz
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (A)
The molarity (M) is given by the formula:
M
On substituting the values in the above equation:
0.02M =
n = 0.002 mol
The number of molecules can be calculated as, number of moles
=
On substituting the values in the above equation:
0.002 mol =
number of molecules = 0.002 6.022 1023
= 12.044 1020
New answer posted
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans:
(i) Hund's Rule (c) Pairing of electrons in the orbitals belonging to the same subshell does not take place until each
orbital is singly occupied.
(ii) Aufbau Principle (e) In the ground state of atoms, orbitals are filled in the order of their increasing energies.
(iii)
New answer posted
10 months agoContributor-Level 10
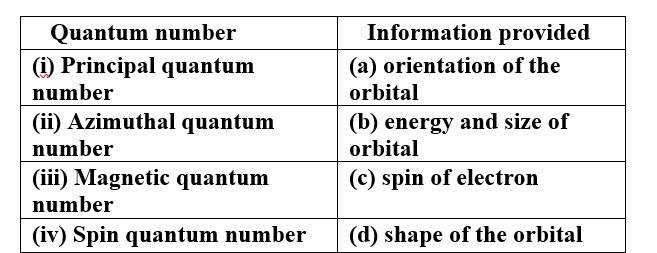
This is a Matching Type Questions as classified in NCERT Exemplar
Ans:
(i) Principal quantum number (b) energy and size of orbital
(ii) Azimuthal quantum number (d) shape of the orbital
(iii) Magnetic quantum number (a) orientation of the orbital
(iv) Spin quantum number (c) spin of electron
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (D)
The number of moles is given by the following formula,
Moles = …. (1)
The number of moles of HCl is calculated by using equation (1) as follows,
Moles of HCl 0.5 mol
The molality (m) is given by the formula:
m =
On substituting the values in the above equation:
Molality = = 1m
New answer posted
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans:
(i) Cu (c) 1s2 2s2 2p6 3s2 3p6 3d10 4s1
(ii) Cu2+ (d) 1s2 2s2 2p6 3s2 3p6 3d9
(iii) Zn2+ (a)1s2 2s2 2p6 3s2 3p6 3d10
(iv) Cr3+ (e) 1s2 2s2 2p6 3s2 3p6 3d3
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers