Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (C)
The molar mass of glucose is 180 g mol- . The molarity (M) is given by the formula:
M =
On substituting the value in the above equation, the mol can be calculated as
M = = 0.005M
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (D)
(A) The number of moles is given by the following formula,
Moles = ….(1)
The number of moles of He is calculated by using equation (1) as follows
Moles of O2 = =1 mol
The number of atoms can be calculated as, number of moles
….(2)
On substituting the values in the above equation:
1 mol =
Number of atoms = 1 6.022 1023
(B) The number of moles of Na is calculated by using equation (1) as follows,
Moles of Na = = 2 mol
The number of atoms can be calculated by using equation (2) as follows,
2 mol =
number of atoms= 2 6.022 1023
(C) The n
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
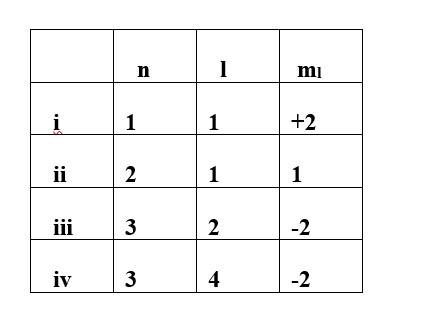
Ans: option (i)& (iv)
- Principal quantum number (n): It represents the size and energy of orbitals.
- Azimuthal quantum number (l): It represents the subshell and shape of orbitals.
- Magnetic quantum number (ml): It represents the orientation of the orbitals.
- Spin quantum number (ms): It represents the direction of spin of electrons in the orbitals.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (B)
The relation between molarity and volume is given as,
M1 V1= M2 V2
On substituting the value in the above equation, the political can be calculated as
5M* 500 mL = M2*1500 mL
M =1.66M
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: option (i) & (iii)
Isoelectronic species are those species that possess the same number of electrons. Here both Na+, Mg2+ possess 10 electrons and both Na+, O2– possess 10 electrons.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: option (ii) & (iii)
For the given value of n (principal quantum number) the value of l (Azimuthal quantum number) varies from 0 to n-1. However for the given value of l the ml (magnetic quantum number) varies from -1 to +1.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: option (i) & (iv)
In a multielectron atomic system the energy of an electron depends not only on its principal quantum number (shell), but also on its azimuthal quantum number (subshell). Electrons having the same shells and same subshells have the same energy and they are known as degenerate orbitals.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (C)
No. of mole given =
On substituting the value in the above equation, the cal, can be calculated as
no, of mole = = 0.1 g
The molarity (M) is given by the formula:
M =
On substituting the values in the above equation:
Molarity =
= 0.2 mol L-1
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: option (iii) & (iv)
The isotopes are defined as atoms with identical atomic numbers but different mass numbers are known as isotopes.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: option (ii) Alpha particle (He2+)
As the wavelength is inversely proportional to the mass of the particles thus the alpha particles would possess the shortest wavelength among the above given option
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers