Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New question posted
11 months agoNew answer posted
11 months agoContributor-Level 10
Benzene is a rich source of electrons because of the presence of an electron cloud containing 6 n-electrons above and below the plane of the ring. Consequently, it attracts the electrophiles (electron-deficient) reagents towards it and repels nucleophiles (electron- rich) reagents. As a result, benzene undergoes electrophilic substitution reactions easily and nucleophilic substitutions with difficulty.
New answer posted
11 months agoContributor-Level 10
3.44. (d) Assertion is a wrong statement. Non-metallic elements have a strong tendency to gain electrons. Therefore, electronegativity is directly related to those non-metallic properties of elements. It can be further extended to say that the electronegativity is inversely related to the metallic properties of elements.
Thus, the increase in electronegativities across a period is accompanied by an increase in non-metallic properties (or a decrease in metallic properties) of elements
New answer posted
11 months agoContributor-Level 10
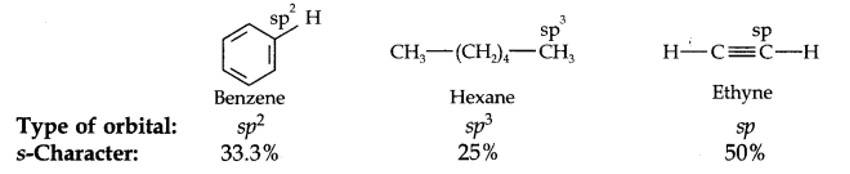
The hybridization state of carbon in these three compounds is:
New answer posted
11 months agoContributor-Level 10
o-Xylene may be regarded as a resonance hybrid of the following two Kekule structures. Ozonolysis of each one of these gives two products as shown below:
New answer posted
11 months agoContributor-Level 10
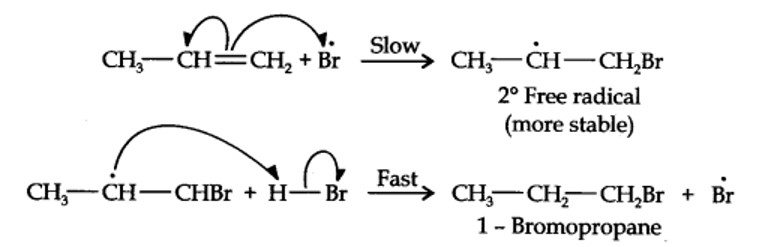
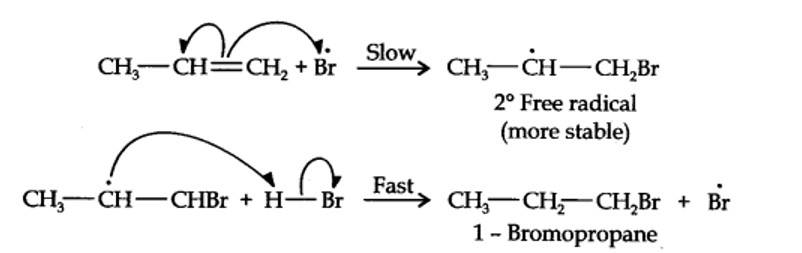
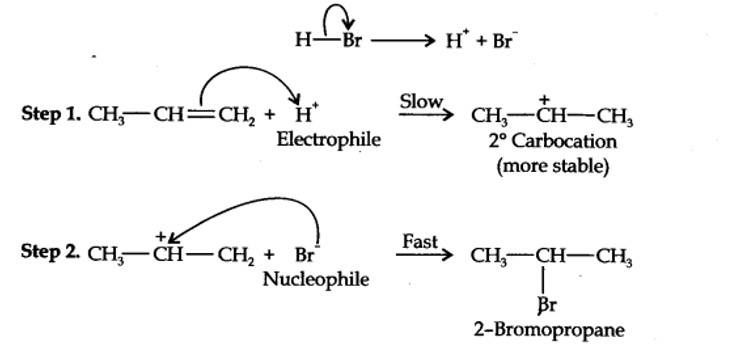
Addition of HBr to propene is an ionic electrophilic addition reaction in which the electrophile, i.e., H+ first adds to give a more stable 2° carbocation. In the 2nd step, the carbocation is rapidly attacked by the nucleophile Br~ ion to give 2-bromopropane.
New answer posted
11 months agoContributor-Level 10
Branching of carbon atom chain decreases the boiling point of alkane.
New question posted
11 months agoNew answer posted
11 months agoContributor-Level 10
3.42. (a) Elements of the same group have similar valence shell electronic configuration and, therefore, exhibit similar chemical properties. However, the elements of the same period have an incrementally increasing number of electrons from left to right, and, therefore, have different valences.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers