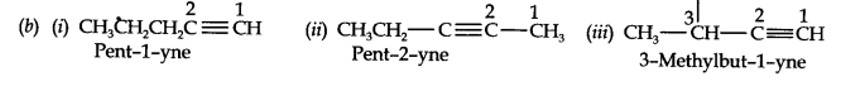Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
3.39. In a period, the non-metallic character increases from left to right. Thus, among B, C, N and F, non-metallic character decreases in the order: F > N > C > B. However, within a group, non-metallic character decreases from top to bottom. Thus, C is more non-metallic than Si. Therefore, the correct sequence of decreasing non-metallic character is: F > N > C > B > Si, i.e., option (c) is correct.
New answer posted
11 months agoContributor-Level 10
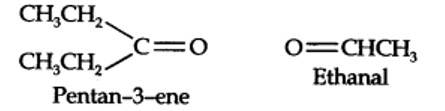
(i) An aldehyde with molar mass of 44 u is ethanal, CH3CH=0
(ii) Write two moles of ethanal side by side with their oxygen atoms pointing towards each other.
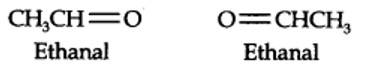
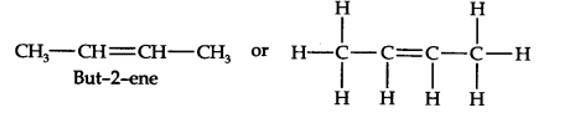
New answer posted
11 months agoContributor-Level 10
3.38. In a period, metallic character decreases as we move from left to right. Therefore, metallic character of K, Mg and Al decreases in the order: K > Mg > Al. However, within a group, the metallic character, increases from top to bottom. Thus, Al is more metallic than B. Therefore, the correct sequence of decreasing metallic character is: K > Mg > Al > B, i.e., option (d) is correct
New answer posted
11 months agoContributor-Level 10
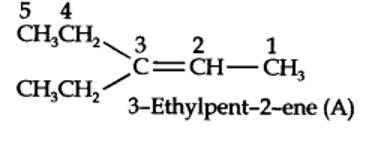
Step 1. Write the structure of the products side by side with their oxygen atoms pointing towards each other.
New question posted
11 months agoNew answer posted
11 months agoContributor-Level 10
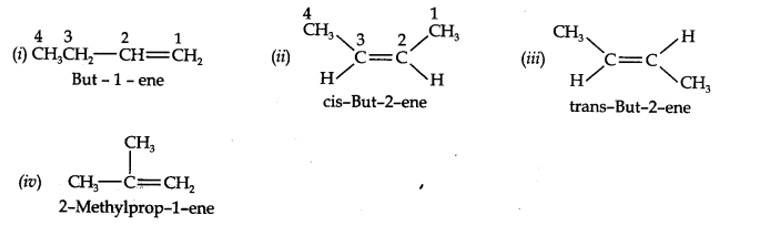
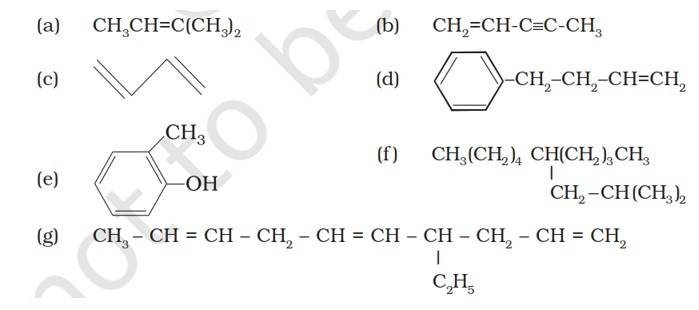
(a) 2-Methykbuut-2-ene
(b) Pent-1-ene-3-yne
(c) But-1,3-diene
(d) 4-Phenylbut-1-ene
(e) 2-Methyl phenol (f) 5- (2-Methylpropyl)decane
(g) 4-Ethyldeca-1,5,8-triene
New answer posted
11 months agoContributor-Level 10
Chlorination of methane is a free radical reaction which occurs by the following mechanism involving initiation, propagation and termination steps:

New answer posted
11 months agoContributor-Level 10
(a) Eutrophication: When the growth of algae increases in the surface of water, dissolved oxygen in water is reduced. This phenomenon is known as eutrophication. (Due to this growth of fish gets inhibited).
(b) Pneumoconiosis: It is a disease which irritates lungs. It causes scarring or fibrosis of the lung.
(c) Photochemical smog: Photochemical smog is formed as a result of the photochemical decomposition of nitrogen dioxide and chemical reactions involving hydrocarbons. It takes place during dry warm season in the presence of sunlight. It is oxidising in nature.
(d) C
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers