Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
sin x = 1 – sin2 x
sin x =
draw y = sin x
y =
find their pt. of intersection.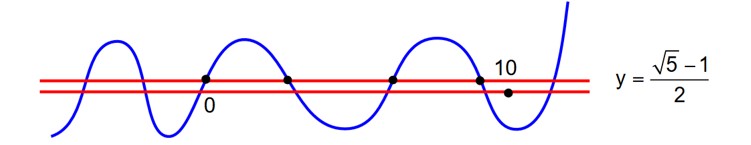
New answer posted
8 months agoNew answer posted
8 months agoContributor-Level 9
Here, meq of MnO2 = meq of Na2S4O6
Mass of MnO2 in sample = 0.261 g
Percentage of MnO2 in sample =
= 13.05%
New answer posted
8 months agoContributor-Level 10
First common term to both AP's is 9
t78 of
t59 of
nth common term
9 + (n – 1) 12 ![]()
n <
Now sum of 19 terms with a = 9, d = 12
New answer posted
8 months agoContributor-Level 9
Pentavalent oxides of group – 15 elements, E2O5 is more acidic than trivalent oxides, E2O3 of the same element.
Acidic strength of trivalent oxides decreases down the group as metallic strength increases.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 690k Reviews
- 1850k Answers