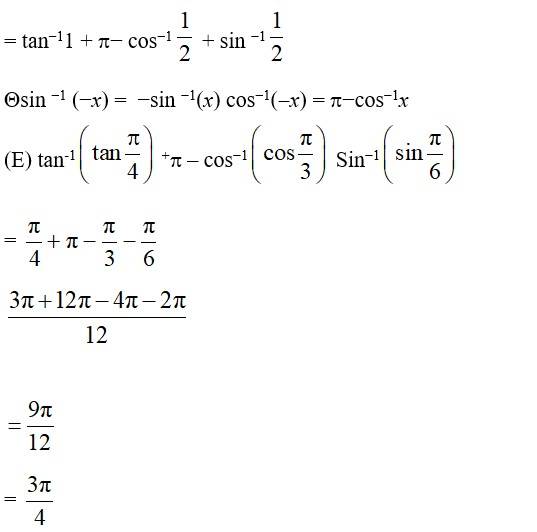Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
Given, Sin−1x=y.
(E) We know that the principal value branch of Sin−1 is
Hence, ≤ y ≤
Option B is correct.
New answer posted
11 months agoContributor-Level 10
Let cos -1 =y Then cos y = = − cos
cos
= cos
= cos
(E) We know that the range of principal value
branch of cos−1 is [0, ] and cos =
Principal value of cos−1 is
New answer posted
11 months agoContributor-Level 10
Let sin−1 =y. Then, sin y=-
We know that the range of principal value branch of sin−1 is
and sin−1
=
Principal value of sin−1
New answer posted
11 months agoContributor-Level 10
4.39 Given, k2 = 4k1, T1 = 293K and T2 = 313K
We know that from the Arrhenius equation, we obtain
On solving, we get,
Ea = 58263.33 J mol-1 or 58.26 kJ mol-1
New answer posted
11 months agoContributor-Level 10
4.38 We know, time t = (2.303/k) * log ( [R]0/ [R])
Where, k- rate constant
[R]0-Initial concentration
[R]-Concentration at time 't'
At 298K, If 10% is completed, then 90% is remaining. t = (2.303/k) * log ( [R]0/0.9 [R]0)
t = (2.303/k) * log (1/0.9) t = 0.1054 / k
At temperature 308K, 25% is completed, 75% is remaining t' = (2.303/k') * log ( [R]0/0.75 [R]0)
t' = (2.303/k') * log (1/0.75) t' = 2.2877 / k'
But, t = t'
0.1054 / k = 2.2877 / k' / k = 2.7296
From Arrhenius equation, we obtain log k2/k1 = (Ea / 2.303 R) * (T2 - T1) / T1T2
Substituting the values,
Ea = 76640.09 J mol-1 or 76.64 kJ mol-1 We know, log k = log A –Ea/RT
Log k =
New answer posted
11 months agoContributor-Level 10
4.37 From Arrhenius equation, we obtain
Also, k1 = 4.5 * 103 s -1
T1 = 273 + 10 = 283 K
k2 = 1.5 * 104 s -1
Ea = 60 kJ mol -1 = 6.0 * 104 J mol -1
Then,
→ 0.5229 = 3133.627 * (T2-283)/ (283 * T2)
→ 0.0472T2 = T2-283 T2 = 297K or T2 = 240 C
New answer posted
11 months agoContributor-Level 10
4.36 We know, The Arrhenius equation is given by k = Ae-Ea/RT Taking natural log on both sides,
Ln k = ln A- (Ea/RT)
Thus, log k = log A - (Ea/2.303RT). eqn 1
The given equation is log k = 14.34 – 1.25 * 104K/T. eqn 2
Comparing 2 equations, Ea/2.303R = 1.25 * 104K
Ea = 1.25 * 104K * 2.303 * 8.314
Ea = 239339.3 J mol-1 (approximately) Ea = 239.34 kJ mol-1
Also, when t1/2 = 256 minutes,
k = 0.693 / t1/2
= 0.693 / 256
= 2.707 * 10-3 min-1 k = 4.51 * 10-5s–1
Substitute k = 4.51 * 10-5s–1 in eqn 2,
log 4.51 * 10-5 s–1 = 14.34 – 1.25 * 104K/T
log (0.654-5) = 14.34– 1.25 * 104K/T = 1.25 * 104/ [ 14.34- log (0.654-5)] T = 668.9K or T =
New answer posted
11 months agoContributor-Level 10
4.35 The given equation is
k = (4.5 x 1011 s-1) e-28000K/T (i)
Comparing, Arrhenius equation
k = Ae -E/RT (ii)
We get, Ea / RT = 28000K / T
⇒Ea = R x 28000K
= 8.314 J K-1mol-1 * 28000 K
= 232792 J mol–1 or 232.792 kJ mol–1
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers