Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
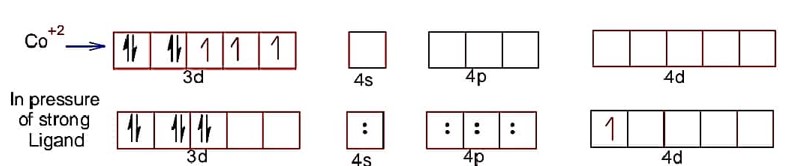
Hence, electronic configuration of CO2+ is
spin magnetic moment =
New answer posted
8 months agoContributor-Level 10
For Ni2+, crystal field CFSE magnitude shows the magnitude of absorbed light. Following are the energy absorbed order
Hence order of colour of compounds are.
New answer posted
8 months agoContributor-Level 10
Both statement are correct for the glass body heating, but reason is not correct explanation during heating process of glass, constituents unit rupture of glass body and gives the edge smoothness.
New answer posted
8 months agoContributor-Level 10
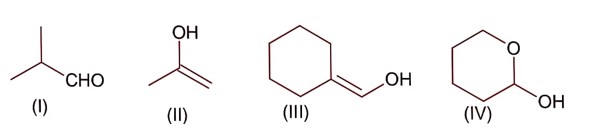
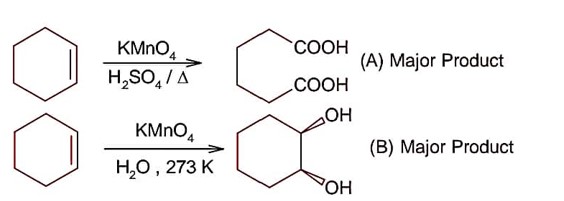
All structure can produce -CHO functional group which gives +ve test of Tollen's reagent except structure (II), because if forms ketonic group after tautomerisation
New answer posted
8 months agoContributor-Level 10
Deep blue colour solution
In cation analysis of Cu+ ions, precipitate formed is CuS on treating with H2S and HCl which dissolved in HNO3 and produced blue colour complex solution
New answer posted
8 months agoContributor-Level 10
Candidates seeking admission to the BTech programme can enrol for admission with Class 12 marks. Candidates must complete Class 12 to enrol for the BTech course. The college offers only one UG course, i.e., BTech. National Fire Service College admissions are based on entrance exam.
New answer posted
8 months agoContributor-Level 10
2nd sentence introduces the subject and gives further information about it in 4th sentence; the use of pronoun 'it' in 1st and 3rd sentence gives further account of the subject.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers