Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
n-factor of KMnO4 in acidic medium = 5
n-factor of Mohr's salt = 1
meq of KMnO4 = meq of Mohr's salt
0.01 * 5 * V = 0.05 * 1 * 20
Volume of KMnO4 used, V = 20 mL
So; Volume of KMnO4 left in burette = 50 -20mL
= 30 mL
New answer posted
8 months agoContributor-Level 10
.For maxima = y = (2n + 1)
For 1st maxima for l1 wavelength (n = 1)
- (1)
First maxima for l2 wavelength
- (2)
New answer posted
8 months agoContributor-Level 10
Moles of C = moles of CO2
=
Mass of C =
= 0.261g
Moles of H = 2 *
Mass of H =
Total mass of compound = 0.492g (given)
So; mass of O = (0.492 – 0.216 – 0.049) g
= 0.227g
% of O =
= 46.14%
the nearest integer = 46
New answer posted
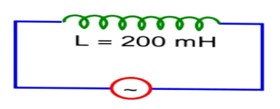
8 months agoContributor-Level 10
Let l = l0 cos wt
Then v = v0 sinwt
at t = 0, v = 0
but l = l0
a = 121 * 2
a = 242
New answer posted
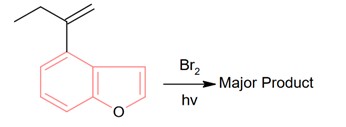
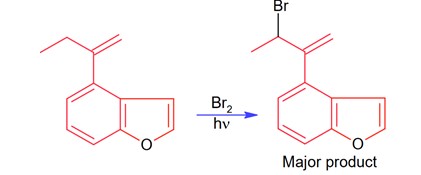
8 months agoContributor-Level 10
Bromination through free radical mechanism occurs at allylic carbon.
New answer posted
8 months agoContributor-Level 10
Ib = 10 µA
IC = 1.5 mA
RL = 50 kW or (Rc)
Base – emitter voltage = 10 mv
=
Av = 750
New answer posted
8 months agoContributor-Level 10
g (x) = px + q
Compare 8 = ap2 …………… (i)
-2 = a (2pq) + bp
0 = aq2 + bq + c
? 4x2 + 6x + 1 = apx2 + bpx + cp + q
? Andhra Pradesh = 4 ……………. (ii)
6 = bp
1 = cp + q
From (i) & (ii), p = 2, q = -1
? b = 3, c = 1, a = 2
f (x) = 2x2 + 3x + 1
f (2) = 8 + 6 + 1 = 15
g (x) = 2x – 1
g (2) = 3
New answer posted
8 months agoContributor-Level 10
(a) will show cis- trans isomerism as;
(b) will not show cis- trans isomerism.
(c) will not show cis- trans isomerism.
(d) will not show cis-trans isomerism.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers