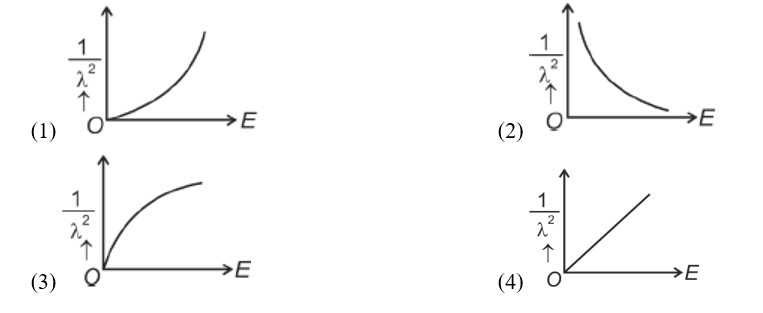Dual Nature of Radiation and Matter
Get insights from 170 questions on Dual Nature of Radiation and Matter, answered by students, alumni, and experts. You may also ask and answer any question you like about Dual Nature of Radiation and Matter
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
Stopping potential defined in terms of wavelength as :
Using above two equation we can calculate :
New answer posted
9 months agoContributor-Level 10
(A) If is the velocity of light
so, (Energy of photon)
(B) Velocity of photon is equal to velocity of light i.e. c.
(C)
(D) In photon-electron collision both total energy and total momentum are conserved.
New answer posted
9 months agoContributor-Level 10
In the case for maximum elongation,
Stress = Elastic limit
i.e. maximum elongation is 4 mm
New answer posted
9 months agoContributor-Level 10
de-Broglie wavelength
where
Squaring both sides,
Graph passes through origin with constant slope.
New answer posted
9 months agoContributor-Level 10
In solar cells, electron emission converts the light energy into electrical energy through the photoelectric effect. Solar cells work due to photoelectric effect, in which photons from sunlight strike solar cell surface. When a photon that has sufficient energy hits the surface, it transfers its energy to an electron in the material of solar cell surface. This causes the electron emission from its bound state. Solar cells can use photoelectric effect to generate electron-hole pairs that are separated and collected for producing electrical current. This provides a renewable source of power.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 690k Reviews
- 1850k Answers